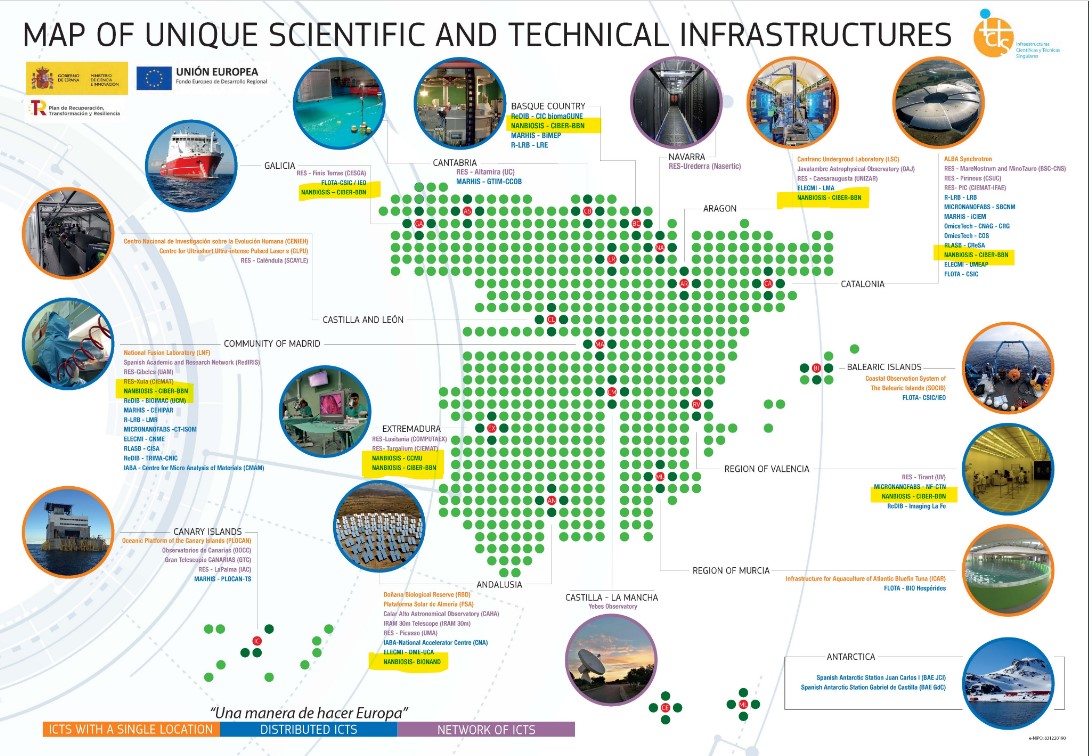
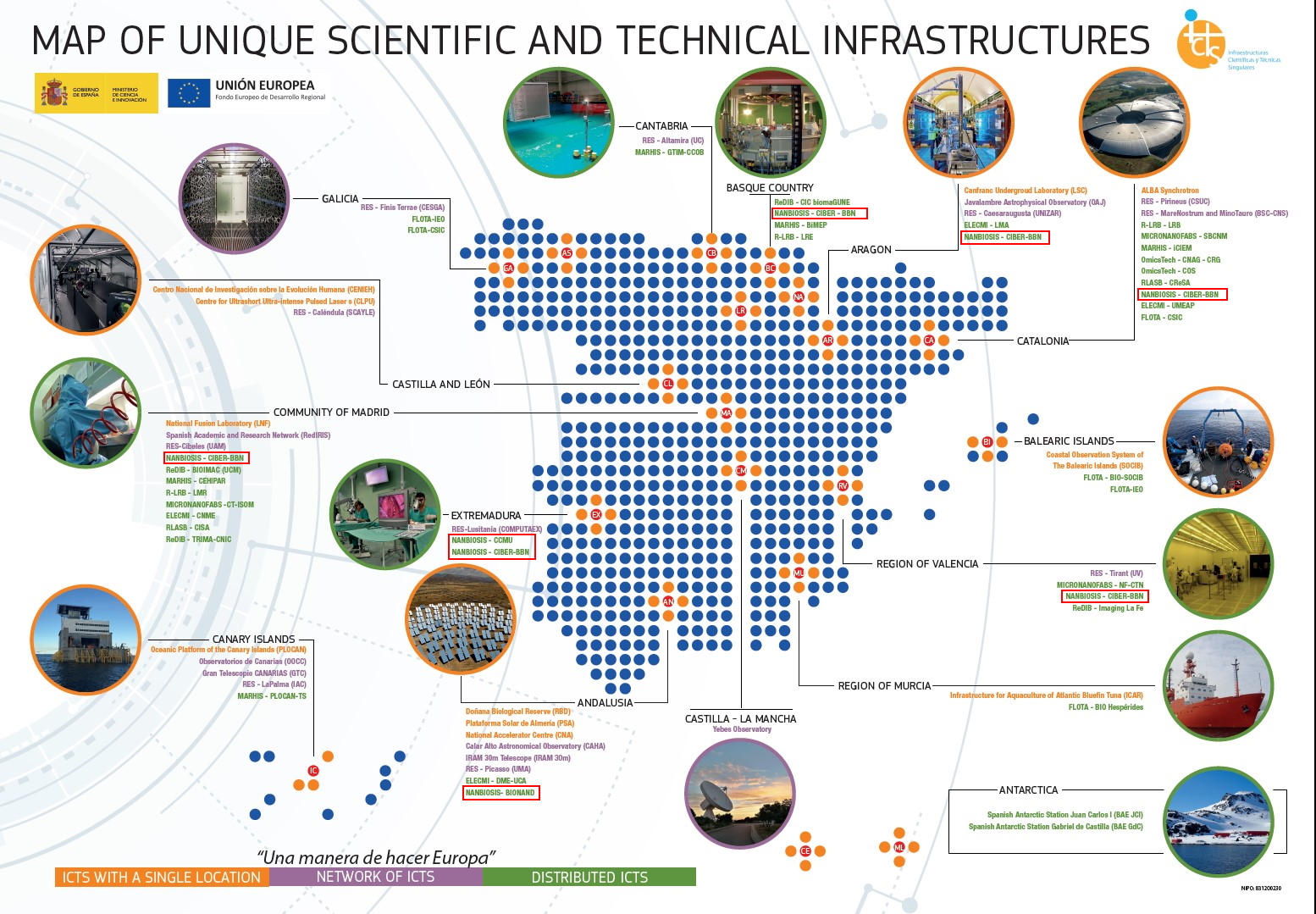
2023 Annual Conference of CIBER-BBN and NANBIOSIS with CIBEREHD
CIBER-BBN´s and NANBIOSIS 2023 Annual Conference will be celebrated in November 6-8, 2023 in Santander
This year we change the format of our annual conferences towards a collective event scheme between the CIBER-BBN and CIBER-EHD thematic areas. Our objective is to create a two-day conference that stimulates scientific collaborations and explores new lines of research. Both areas have already shared experiences of collaborative projects, demonstrating the complementarity of their fields. The results of these seed projects will be presented on the first day. In addition, we are pleased to announce that there will be a new edition of seed collaborative projects between the two areas, which will open in the autumn, and close after the joint meeting.
The anual session dedicated to NANBIOSIS is scheduled for the afternoon of the Tuesday 7th, After that, we will cellebrate the annual meeting of the NANBIOSIS coordination Committee and the Scientific and Technical Advisory Board of NANBIOSIS.
We hope that you find the program attractive and that we can enjoy a few days of science, friendship, community, collaboration and discovery.
Click here to view the program and register
Related news