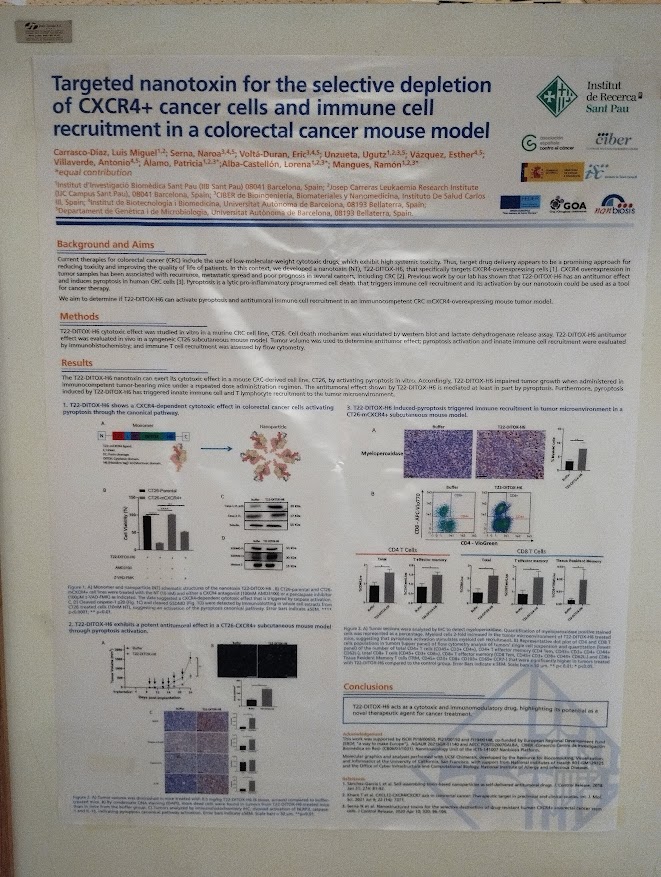
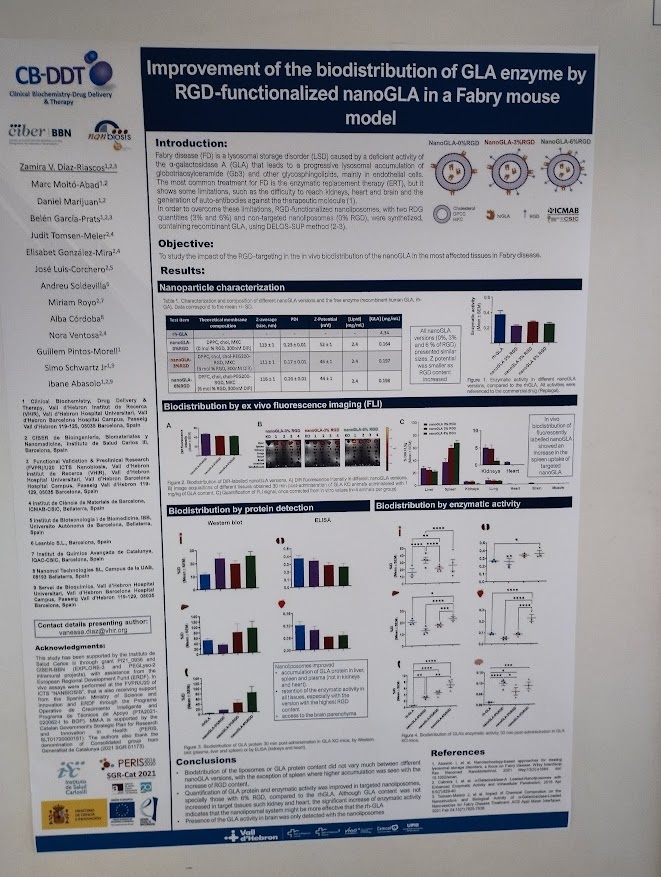
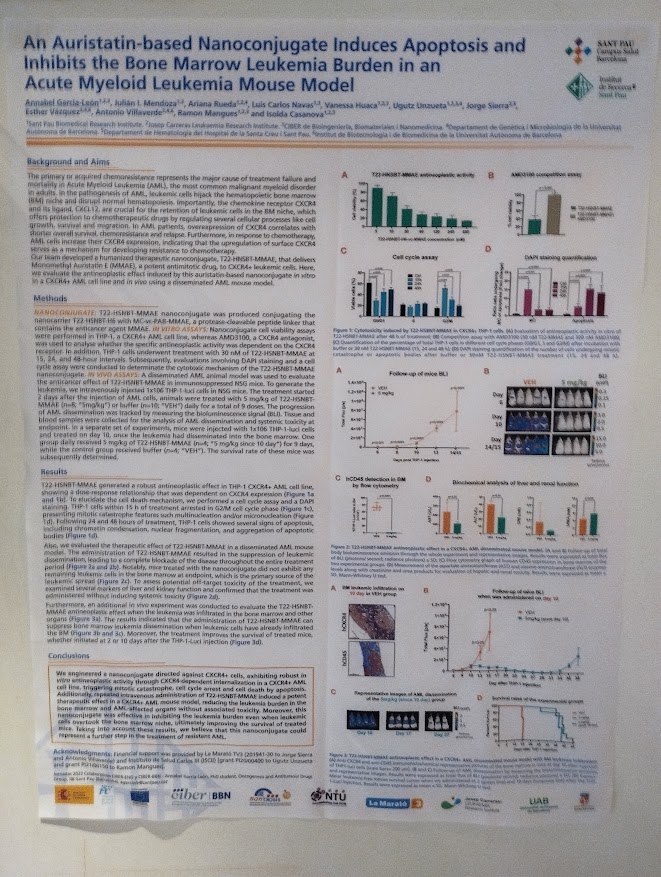
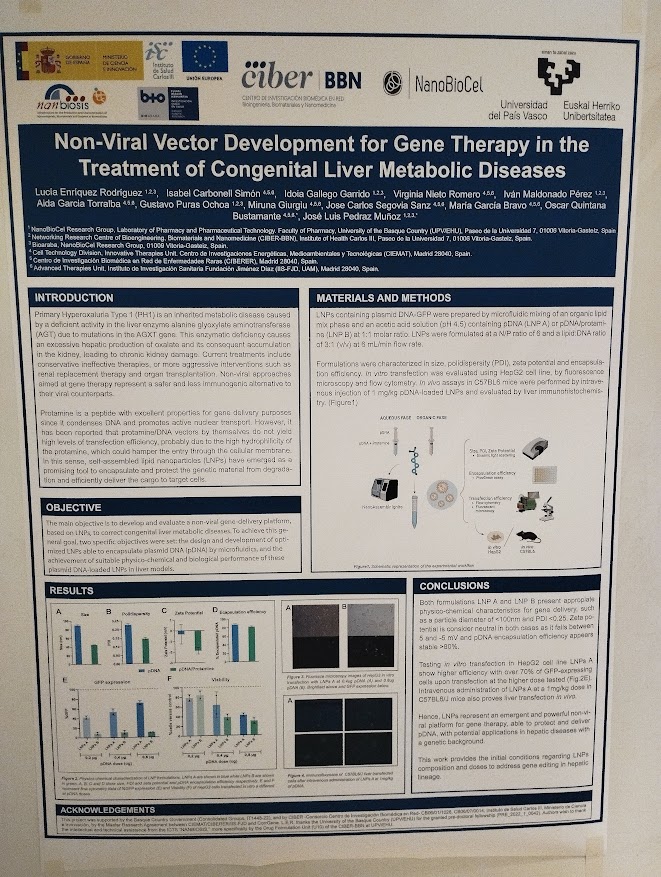
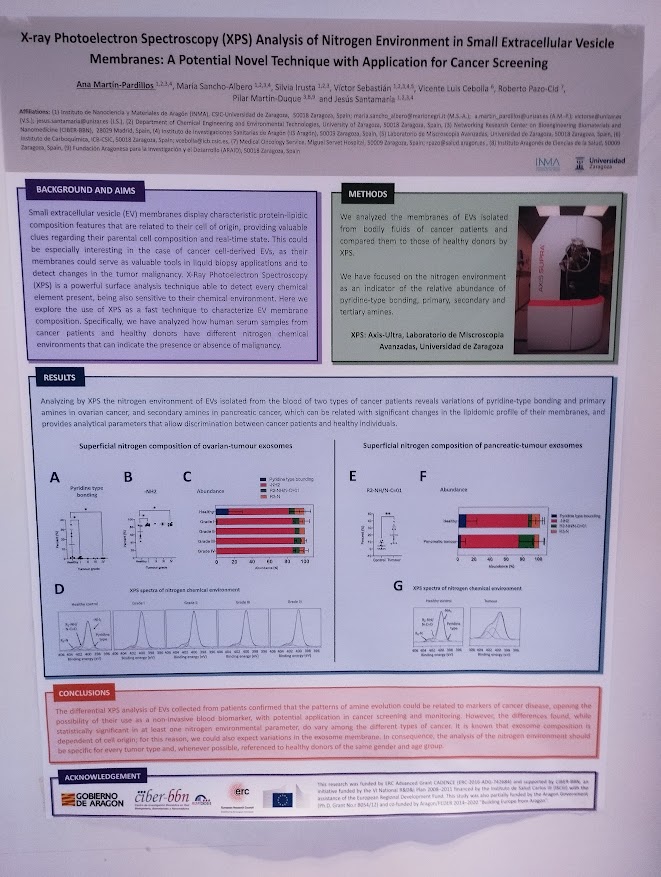
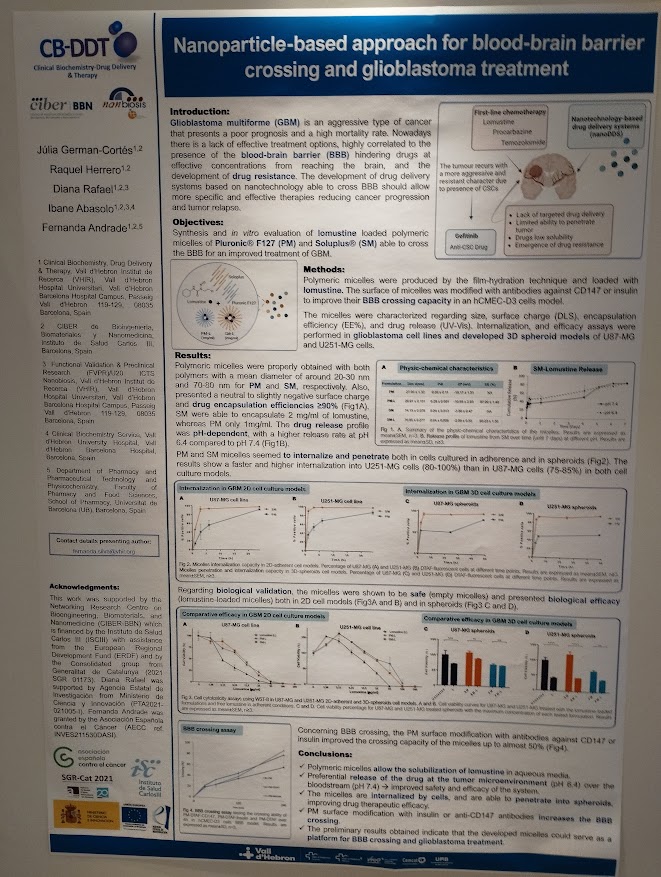
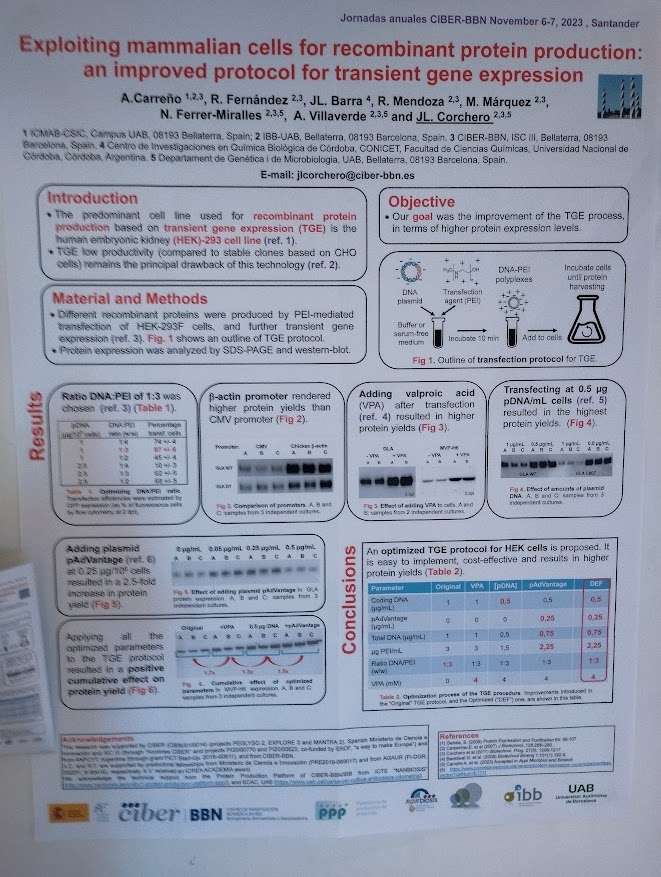
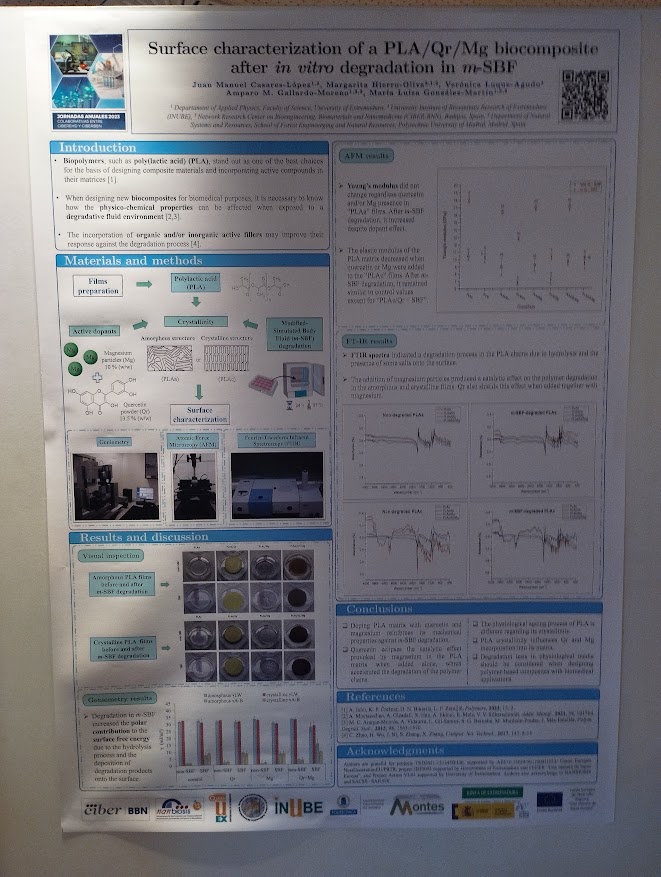
Rosa Hernández of the NanoBioCel group-NANBIOSIS U10 has received the Female Talent Award 2023 Women and Science
On Thursday, November 2, the Female Talent Award, in the category of Women and Science, was awarded to Rosa Hernández from the NanoBioCel group of CIBER BBN and the U10 Drug Formulation Unit. The award is given by AMPEA (Association of Professional and Business Women of Alava).
In her award speech, Rosa Hernández, highlighted that the effort and work carried out during all these years has allowed the research group to develop cutting-edge research and to place the names of the University of the Basque Country (UPV/EHU) and Vitoria-Gasteiz on the map of cutting-edge research in Nanotechnology and Regenerative Medicine.
She also stressed that: “awards like this allow society to get to know us, to know what we do in our laboratories and gives us a lot of visibility so that girls and young women begin to have female references in science and will be encouraged to choose, in the future, a scientific career”.