U2. Custom Antibody Service Unit (CAbS)
The facility is located in the Institute of Advanced Chemistry of Catalonia (IQAC-CSIC; Barcelona). It is equipped with a laboratory for the development and production of monoclonal antibodies. The laboratory is fully equipped for obtaining, selecting and storing monoclonal antibody producing hybridomas. In addition, the facility has infrastructure for the production of polyclonal antibodies, as well as laboratories for the synthesis of bioconjugates, haptens, the purification of antibodies and the performance of immunoassays. In addition to the above mentioned equipment, the IQAC-CSIC provides animal husbandry facilities and a unit for the synthesis of organic molecules. The equipment consists in an inverted phase contrast microscope with a camera, two Class II laminar flow biological safety cabinets, two CO2 incubators, a bioreactor for hybridoma culture, and tangential filtration unit for purification of the antibodies produced in the culture. Among the items of available equipment it should be highlighted a spotter for selection of high antibody producing cell lines and an automated ELISA microplate washer an ELISA reader, two AKTA Prime Liquid chromatography systems for antibody purification, plus standard equipment (centrifuges, fridges, liquid nitrogen containers, autoclave, baths, and vacuum equipment) and all the necessary routine laboratory apparatus.Cabs also has access to a fully equipped chemistry laboratory for the synthesis of haptens and immunoreagents. FOR THOSE SERVICES IDENTIFIED AS OUTSTANDING, AT LEAST 20% OF THEIR CAPACITY IS OPEN UNDER COMPETITIVE ACCESS. SEE ANNEX 1 OF ACCESS PROTOCOL FOR DETAILS ON % OF OPENNESS FOR EACH SERVICE
28 Feb Researchers from IQAC-CSIC advance towards faster detection and treatment of cystic fibrosis and rare respiratory diseases, improving patient outcomes. 28 February 2024, IQAC-CSIC (Barcelona) Cystic Fibrosis (CF) is a progressive autosomal recessive disease. It is caused by a mutation in the gene encoding the Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) protein, disrupting its exocrine activity. While CF affects various organs, its impact on the lungs is particularly severe. This disease leads to the accumulation of thick, sticky mucus, obstructing airways and trapping bacteria, resulting in significant infections and extensive lung damage. Consequently, individuals with CF are highly susceptible to respiratory[...] 24 Nov During 20-22 of November 2023, the III PTI+Global Health Scientific Conference were held in the Center for Human and Social Sciences, in Madrid. In March 2020, the CSIC (Spanish National Research Council) launched the the Interdisciplinary Thematic Platform (PTI) on Global Health to bring together research teams and enhance knowledge about the new coronavirus SARS-CoV-2, which caused the pandemic. The PTI has mobilized and coordinates more than 400 scientists from 50 CSIC institutes in all areas. The annual PTI+Global Health Scientific Conference are a meeting space where the results of the research carried out in the laboratories can be shown[...] 08 Nov The annual conference of the scientific áreas of CIBER (the most important Centre for Biomedical Research in Spain) are hotly awaited every year for the CIBER community as a foro to be updated about emerging key technologies and discuss about research lines and results, find new opportunities to collaborate and join efforts towards common objectives. Moreover, this year, the Annual Conference of CIBER-BBN (Bioengineering, Biomaterials and Nanomedicine) has been organized as a collective event scheme together with the scientific area of CIBEREHD (Digestive and Liver Diseases). Both areas have already shared experiences of collaborative projects, demonstrating the complementarity of their fields.[...] 20 Oct Today, Bonnie Bassler, Jeffrey Gordon, and Peter Greenberg are receiving the Princess of Asturias Award for their contribution to the knowledge of the role of the human microbiome and the bacteria Quorum Sensing. Targeting Quorum Sensing allows understanding of bacteria behaviour and pathogenesis, but also developing new therapeutic and diagnostic strategies for infectious diseases. Bonnie Bassler has shown how small chemical signals are involved in such a communication process. Peter Greenberg was one of the first scientists who assigned the term Quorum Sensing (QS) to such a sophisticated system used by bacteria to communicate and synchronize behavior. He was involved[...] 15 May Nb4D has a new look on line! Nb4D Group (of IQAC-CSIC and CIBER-BBN) has lunched a new website “Revolutionsising Diagnosis“ with the aim to facilitate a faster and easier navigation througth their “pioneering research to develop new diagnostic and therapeutic approaches” and their solutions and expertise to help researchers and companies. Antibodies, bioreceptors, hapten design and synthesis, immunoanalytical method development, new ivd tools, surface functionalization, therapeutic antibodies and much more knowledge and expertise revolutionising diagnosis. The new website contains a page for CAbS-NANBIOSIS. Custom Antibody Service (CAbS), unit 2 of the ICTS NANBIOSIS[...] 28 Feb Today is the international day of rare diseases, a day to raise awareness and instigate change for people living with a rare disease. From NANBIOSIS we want to sume to this celebration and higtligh our commitment to helping people with rare diseases through research. Dr. Ibane Abasolo, Scientific Director of NANBIOSIS U20, was at the WORLDSymposia conference last week in Orlando (FL, USA), where the latest advances in preclinical study and clinic of lysosomal storage diseases were explained. There, she presented the results obtained in the Smart4Fabry project coordinated by the CIBER-BBN where nanoliposomes were developed for the treatment of[...] 27 Feb Several CIBER-BBN groups at the University of Barcelona, the Institute for Advanced Chemistry of Catalonia (IQAC-CSIC), the Institute of Microelectronics of Barcelona (IMB-CNM-CSIC) and the Institute of Nanoscience and Materials of Aragon (INMA) —a joint institute of the CSIC and the University of Zaragoza (UNIZAR)— have developed a new method for detecting RNA viruses based on the technology of using probes that form triplex structures. This innovative methodology opens up new options to detect viruses such as SARS-CoV-2, the influenza A (H1N1) virus or the respiratory syncytial virus (RSV), a pathogen that affects newborns and requires differential diagnostic care. This[...] 15 Nov Today, November 15 is a day of celebration for us, the Day of the Chemistry in Spain! Chemistry is the science that studies matter, how it is composed, its properties and how its structures are transformed and, as matter is everything, including living beings and ourselves, we can say that chemistry is omnipresent and transversal in all areas surrounding us. Chemistry is everywhere, we ourselves are chemistry and our health and our life is chemistry. “Everything around us is chemistry in the environment, foods, what we use and what we touch every day. Our own body is a sophisticated complex[...]
U2. Custom Antibody Service (CAbS)



Description
Services
Equipments
Active projects
Other projects
Ref
Title
Funding Organism
Unit Role
SAF2015-67476-R
Immunochemical Strategies of Diagnosis and Therapy based on Quorum Sensing (ImmunoQS)
Ministerio de Economía y Competitividad
Participant
PIE1400061
Molecular links between diabetes and neurodegenerative diseases
ISCIII
Oberver
2014SGR1484
Consolidated Research Group
Generalitat de Catalunya
Participant
FP7-614168
Real time monitoring of SEA contaminants by an autonomous Lab-on-a-Chip biosensor (SEA-on-a-CHIP)
European Commission
Oberver
CI16-00031 Caixa Impulse
A new approach for the detection of Pseudomonas aeruginosa
CaixaBank
Participant
MSCA-ITN2015-675412
New Diagnostics for Infectious Diseases
European Commission
Participant
TECCTP15-1-0011
Ajuda acreditació Tecnio del grup Nb4D
ACCIÓ: Generalitat de Catalunya
Others
Publications
2016
News U2

The fight against rare respiratory diseases: New hope in detection and treatment

From the molecule to the bioassay by Custom antibody service (CAbS)-NANBIOSIS U2 as a PTI+Global Health Infraestructure

NANBIOSIS sesion in the CIBER-BBN and CIBEREHD annual meeting. SAFE-N-MEDTECH Project: Outcomes and Future Prospects.

The importance of bacterial Quorum Sensing (QS) research recognised in the Princesa of Asturias Awards

New look of Nb4D – CAbS (NANBIOSIS U2) “Revolutionising Diagnosis”

Rare diseases international day 2023: some NANBIOSIS contributions
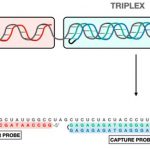
New method for the detection of RNA viruses such as SARS-CoV-2

Happy Day of Chemistry! The role of Chemistry in a sustainable research in health