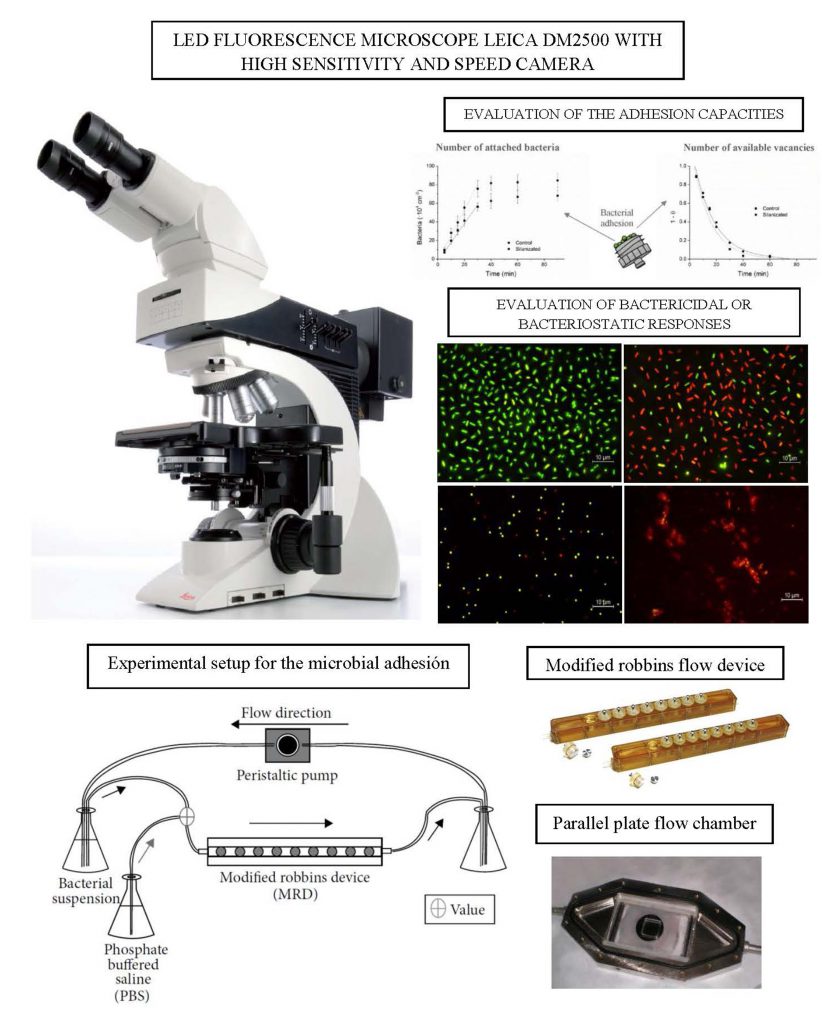
U16-S09. Kelvin Probe Force Microscopy (KPFM) and Atomic Force Microscopy (AFM)
Electrical properties of solid surfaces using Kelvin Probe Force Microscopy (KPFM) and mechanical and topographical quantification using Atomic Force Microscopy (AFM)
The AFM Dimension Icon-PT has a microscopy method that allows to obtain the electrical properties of the surface of a material in air or liquid. In addition, the mechanical and viscoelastic properties of these surfaces can be determined and their morphology and topography can be studied qualitatively and quantitatively through images and 3D roughness parameters. The measuring range of this equipment reaches tenths of nanometres in height and hundreds of nanometres in spacing.
Customer benefits
The service will be provided by specialists in characterisation of materials in micro-nanoscopic dimensions.
The service works in accordance with ISO, ASTM, ASME and EUR standards. In addition, it can also provide detailed reports according to the needs of the study.
Target customer
The service can meet the needs of companies by providing support for their ISO, ASTM, ASME and EUR controls. In addition, it can also service research groups with detailed reports according to the needs of the study.
Additional information