NANBIOSIS Unit 6 Research Featured During ICMAB Scientific Advisory Board Visit
NANBIOSIS Unit 6 contributed to advanced nanomedicine research showcased at the ICMAB Scientific Advisory Board visit.
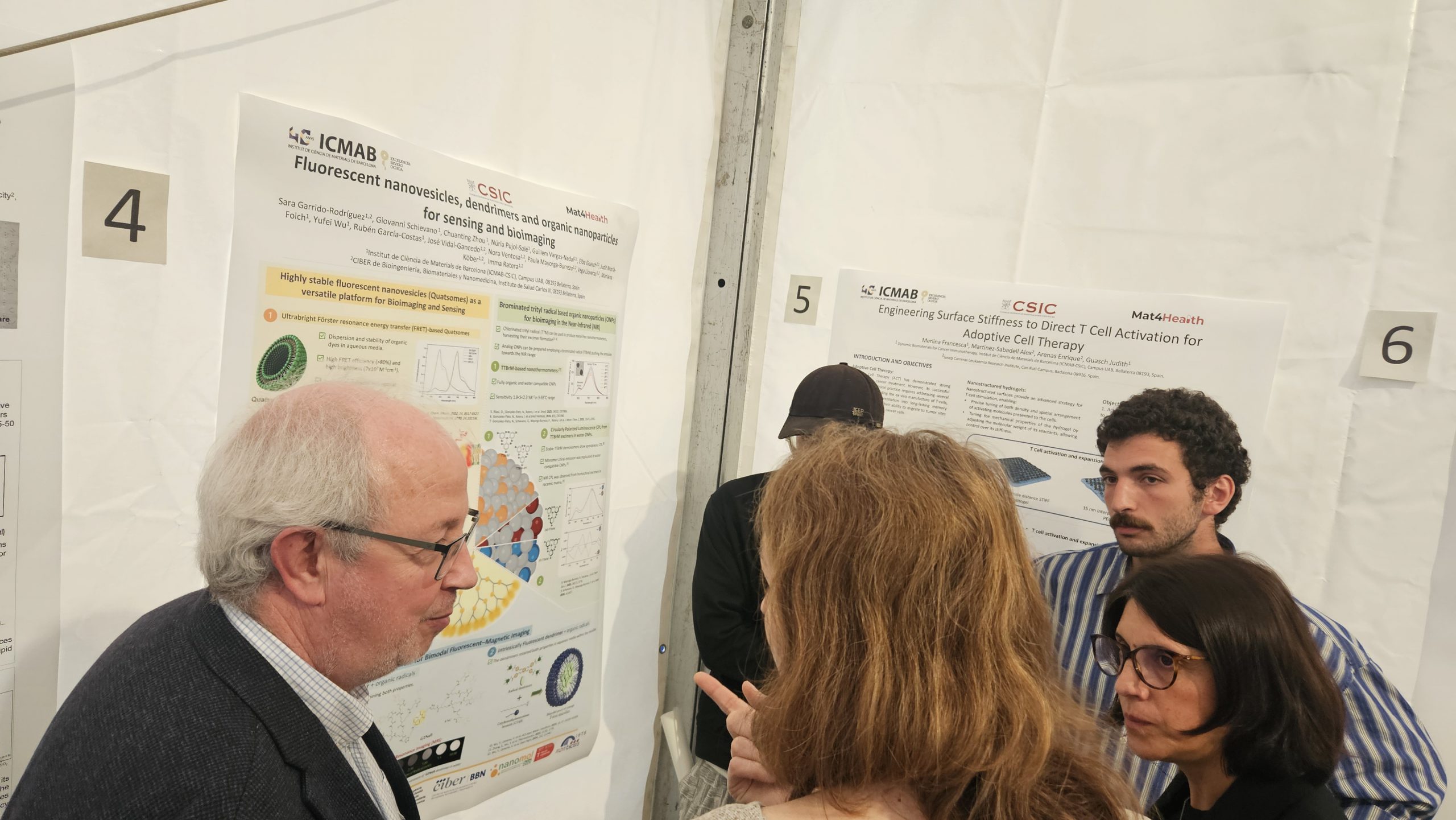
Barcelona, May 2026 — Researchers from Institut de Ciència de Materials de Barcelona (ICMAB-CSIC) recently presented a poster highlighting innovative developments in fluorescent nanovesicles, dendrimers, and organic nanoparticles for sensing and bioimaging during the visit of the institute’s Scientific Advisory Board (SAB). The visit formed part of the evaluation activities linked to the Severo Ochoa excellence accreditation awarded to ICMAB.
The poster showcased multidisciplinary research focused on the development of advanced nanomaterials for biomedical imaging, biosensing, and diagnostic applications. Among the technologies presented were highly stable fluorescent quatsomes for bioimaging and nucleic acid sensing, ultrabright nanoparticles for deep-tissue imaging, and multifunctional dendrimers combining fluorescent and magnetic resonance imaging (MRI) capabilities.
Part of this research was carried out with the support of NANBIOSIS Unit 6, whose contribution was acknowledged in the poster. The Scientific Director of NANBIOSIS, Ramón Martínez Máñez, also participated in the Scientific Advisory Board committee involved in the visit.
The work reflects the growing impact of nanotechnology and advanced biomaterials in the development of next-generation biomedical imaging and sensing tools. These innovative platforms could contribute to improved diagnostics, real-time monitoring, and future personalized medicine applications.
The poster included contributions from researchers affiliated with CIBER-BBN and ICMAB-CSIC, with several of the presented studies recently published in leading scientific journals in materials science and nanomedicine.
NANBIOSIS Unit 6: Advanced materials for health
The poster highlights the mission of NANBIOSIS Unit 6, located at ICMAB-CSIC and coordinated by Prof. Nora Ventosa. This Unit specializes in the development, characterisation, and scale-up of molecular biomaterials with applications in biomedicine.
Equipped with state-of-the-art facilities and operating under ISO 9001 quality standards, Unit 6 offers advanced technologies such as the DELOS-SUSP method, a compressed-fluid-based process for producing highly controlled micro- and nanostructured materials. Its unique capability to scale processes from laboratory to industrial levels supports both research and technology transfer.
What is NANBIOSIS?
The goal of NANBIOSIS is to provide comprehensive and integrated advanced solutions for companies and research institutions in biomedical applications. All of this is done through a single-entry point, involving the design and production of biomaterials, nanomaterials, and their nanoconjugates. This includes their characterization from physical-chemical, functional, toxicological, and biological perspectives (preclinical validation).
If you want to collaborate with us, visit our Order Request page.
In order to access our Cutting-Edge Biomedical Solutions with priority access, enter our Competitive Call here.
NANBIOSIS has worked with pharmaceutical companies of all sizes in the areas of drug delivery, biomaterials and regenerative medicine. Here are a few of them: