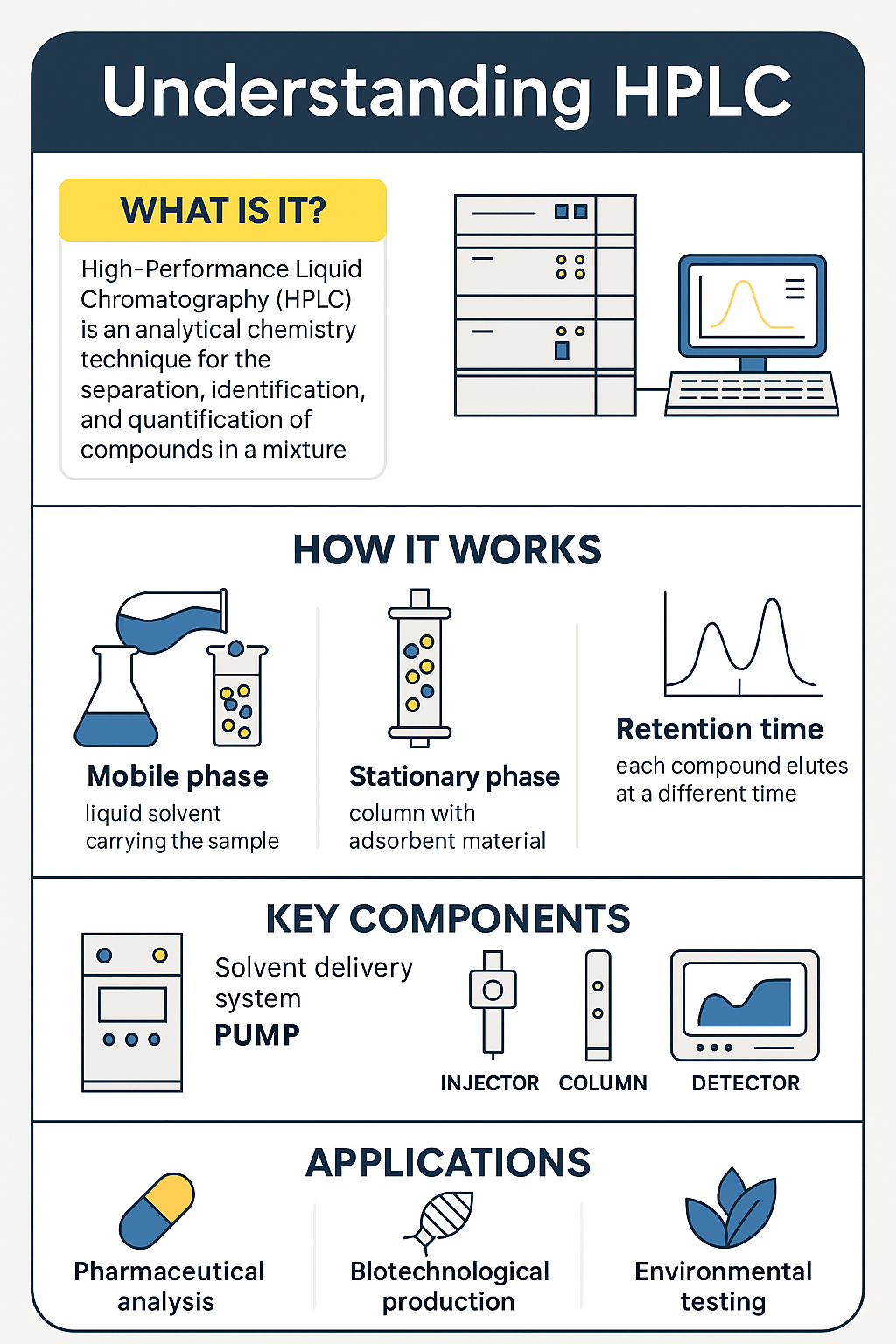
Biofunctionalized nanoconjugates: Advancing diagnostics and nanomedicine
NANBIOSIS launches an integrated service to develop antibody-functionalized nanoconjugates, bridging diagnostics and nanomedicine innovation.
NANBIOSIS introduces a cutting-edge integrated service for the design, production, and characterization of biofunctionalized nanoconjugates, addressing a critical bottleneck in diagnostics and nanomedicine development.
Led by Prof. M. Pilar Marco (Unit 2, CAbS) and Dr. Carlos Rodríguez (Unit 12), and coordinated by Nuria Pascual (U2) and Susana Vilchez (U12), this initiative combines expertise across disciplines to deliver robust, reproducible, and application-ready nanobiotechnology solutions.
Bridging a Critical Gap in Nanomedicine
Many promising biomedical innovations fail to progress beyond the prototype stage due to a disconnect between two key components: the biological recognition element (such as antibodies) and the nanostructured carrier.
Developing these elements independently often leads to incompatibilities in performance, stability, and scalability—especially in advanced applications such as:
- Antibody-functionalized nanoprobes
- Liposomal or micellar diagnostic reagents
- Point-of-care nanodiagnostics and theranostics
NANBIOSIS addresses this challenge by integrating both aspects into a single, coordinated workflow.
A Fully Integrated Workflow
This new service connects the strengths of Unit 2 (Custom Antibody Service, CAbS) and Unit 12 (Nanostructured Liquids Characterization Unit), offering a seamless pipeline from concept to validated nanoformulation.
Key capabilities include:
- Custom antibody development (U2):
Design of haptens and peptides, antibody production and purification, immunoassay development, and bioconjugation. - Advanced nanomaterial characterization (U12):
Comprehensive physicochemical analysis of colloidal systems, including size distribution, zeta potential, stability, viscosity, and microscopy (e.g., DLS/SLS and SEM).
Together, these services enable the development of optimized antibody-functionalized nanoconjugates with enhanced robustness and translational potential.
From Concept to Application
The integrated workflow covers all stages of development:
- Target definition and biomarker selection
- Hapten and immunogen synthesis
- Antibody production and screening
- Functionalization of nanostructures
- Physicochemical characterization
- Stability and performance optimization
This end-to-end approach ensures compatibility between biological and nanostructured components, significantly increasing the likelihood of successful real-world applications.
Proven Impact in Biomedical Research
The service builds on successful collaborations and high-impact research. Notable examples include:
- A nanoplasmonic biosensor for monitoring the anticoagulant acenocoumarol in plasma, integrating highly specific antibodies with gold nanostructures.
- The development of functionalized PLGA nanoparticles for theranostic applications, demonstrating selective interaction with proteins involved in transport across the blood–brain barrier.
These cases highlight the potential of integrated nanobioconjugation strategies to advance precision diagnostics and targeted therapies.
Enabling the Future of Nanodiagnostics
By combining biological expertise with advanced nanomaterial characterization, NANBIOSIS positions itself at the forefront of nanomedicine, biosensing, and point-of-care diagnostics.
This integrated service offers researchers and companies a reliable pathway to transform innovative ideas into validated, high-performance nanobiomedical products—accelerating their journey from lab to market.
You can read more information about this CEBS here.
What is NANBIOSIS?
The goal of NANBIOSIS is to provide comprehensive and integrated advanced solutions for companies and research institutions in biomedical applications. All of this is done through a single-entry point, involving the design and production of biomaterials, nanomaterials, and their nanoconjugates. This includes their characterization from physical-chemical, functional, toxicological, and biological perspectives (preclinical validation).
If you want to collaborate with us, visit our Order Request page.
In order to access our Cutting-Edge Biomedical Solutions with priority access, enter our Competitive Call here.
NANBIOSIS has worked with pharmaceutical companies of all sizes in the areas of drug delivery, biomaterials and regenerative medicine. Here are a few of them: