Lucía Enríquez at Kyoto University: Advancing Cystic Fibrosis Research Through iPS Technology
Lucía Enríquez shares her research stay at Kyoto University, advancing cystic fibrosis models with iPS cells and NANBIOSIS Unit 10 technologies.
Kyoto, February 2026 — International research stays are key to accelerating innovation in biomedicine. Lucía Enríquez, a PhD researcher linked to NANBIOSIS Unit 10, is currently carrying out a research stay at Kyoto University, one of the world’s leading institutions in stem cell research. In this interview, she shares how this experience is helping bridge advanced induced pluripotent stem cell (iPS) technology, non-viral gene delivery systems, and 3D bioprinting to improve disease modeling for cystic fibrosis.
Below is the full interview transcript. See the video at the end of this article.
Interview with Lucía Enríquez
Q: Lucía, you are currently carrying out a research stay in Kyoto. Where are you working, and why is this center considered a world leader in its field?
Answer:
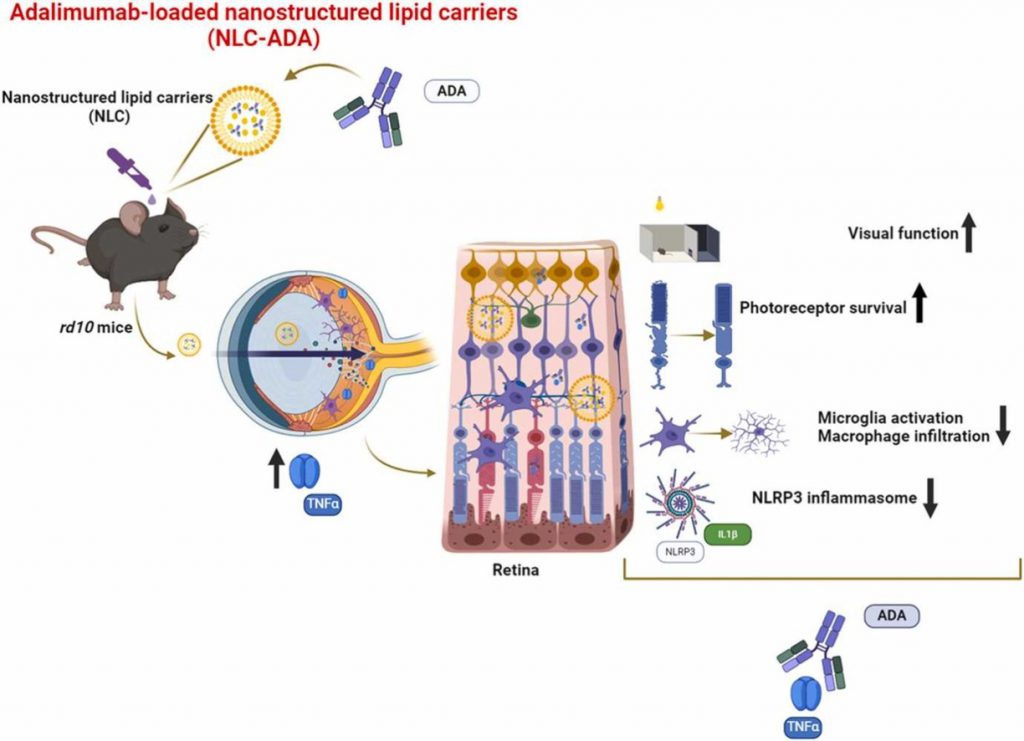
Right now, I’m working at Kyoto University. I’m doing an international research stay during my PhD in Spain, which is focused on studying cystic fibrosis as a pathology. What we’re doing here are models of the disease that are more representative of the patient’s genetic profile.
For this purpose, we’re using iPS cells, which are cells derived from patients that are reprogrammed to be pluripotent. iPS stands for Induced Pluripotent Stem cells. What we do is obtain cells from a patient, reprogram them to be pluripotent, and then differentiate them into the tissue that we want to study. In this case, since cystic fibrosis has a strong impact on the pulmonary system, we develop them into airway epithelium.
The reason why this center is a leader in the field is because it was founded by Shinya Yamanaka, who received the Nobel Prize in Physiology or Medicine in 2012. He discovered the reprogramming factors that allow a fully differentiated cell to be transformed back into a pluripotent state — an induced pluripotent stem cell.
Q: This technology seems very useful in personalized medicine. Cystic fibrosis is considered a rare disease, correct?
Answer:
Yes, cystic fibrosis is one of the least rare among rare diseases, but it is still considered a rare disease. And yes, iPS technology helps you model genetic pathologies very effectively.
Studying human genetic diseases is complex because traditional in vitro cellular models usually involve only one cell type. That does not reflect physiological reality. With iPS cells, you can generate multicellular structures in vitro derived from the same patient, meaning they are genetically homogeneous. This allows you to better understand what is happening and how cells respond, in a more physiologically relevant environment — without relying directly on human or animal models.
Q: You obtained a JSPS fellowship, which is highly competitive. What did this opportunity mean to you?
Answer:
The JSPS is the Japan Society for the Promotion of Science, which offers several grants. I was awarded a short-term postdoctoral fellowship. It is primarily intended for international postdoctoral researchers, but it can exceptionally be awarded to final-year PhD students with strong projects.
They award 25 grants per call worldwide, so it is highly competitive. I was fortunate to be one of the non-postdoctoral exceptions.
Professionally and personally, it has been a great experience. Moving from Spain to Japan and immersing myself in a different research and cultural environment has been enriching. Importantly, this grant included research funding — the first time I have had dedicated funding to manage myself. This allowed me to attend a Keystone symposium in Kyoto in January, which would not have been possible otherwise.
Q: How does this research connect with your PhD work at NANBIOSIS Unit 10?
Answer:
At NANBIOSIS Unit 10, we have two main lines of work:
- The synthesis, optimization, production, and characterization of non-viral vectors for gene delivery and other delivery systems.
- 3D bioprinting and characterization of human tissues, mainly for regenerative medicine and disease modeling.
The idea was to combine these technologies: iPS cell technology from Kyoto, non-viral gene delivery systems, and 3D bioprinted airway epithelia modeling cystic fibrosis tissue.
We aim to develop non-viral vectors to transfect or treat iPS cells and use them to generate 3D bioprinted models of cystic fibrosis airway epithelium.
Q: What new approaches have you found in this laboratory compared to Spain?
Answer:
I have not found dramatic differences in research workflows, which is actually reassuring.
I came here specifically to learn about iPS technology and differentiation into pulmonary lineage cells. These cells behave differently from immortalized cell lines. They are more sensitive and require different characterization approaches.
I have learned new workflows that complement my previous experience in Spain. The goal is to merge this new knowledge with what we do at home and strengthen collaborative approaches.
Q: How would you describe the capabilities and international position of NANBIOSIS Unit 10?
Answer:
In science, you rely on experts with different skill sets. Good research centers have strong infrastructures and specialized technical staff.
NANBIOSIS is a platform that allows researchers to expand their ideas beyond a single center, at a national level. It is a very unique infrastructure. Here in Japan, services are often internal to each research center. There is less of a nationwide shared infrastructure model.
With NANBIOSIS, researchers can access a wide range of expertise across Spain. I believe this is something we should value and promote internationally.
Q: What can you contribute to the Kyoto laboratory?
Answer:
iPS cells are widely used for tissue regeneration and disease modeling.
In regenerative applications, lipid-based non-viral vectors can genetically modify cells before transplantation, increasing therapeutic potential in a safer way.
In disease modeling, these vectors can be optimized in vitro before moving to in vivo studies. These technologies are highly complementary and work very well together.
Q: Has living and working in Kyoto influenced your scientific perspective?
Answer:
Yes. I’m in the final year of my PhD, and this is when you start reflecting on your future.
Being here has broadened my perspective — professionally and personally. It has shown me that opportunities can arise anywhere. In my opinion, very few things are truly impossible. Keep your doors open and work towards what you want.
Q: What advice would you give to young researchers considering international fellowships?
Answer:
If you’re a researcher, you’re probably curious by nature — embrace that curiosity.
Be persistent. Talk to people. You will receive many “no’s,” but that is part of research and life. Don’t be afraid to reach out.
Some people told me not to apply for this grant because it was too competitive. My answer was simple: maybe I won’t get it, but it won’t be because I didn’t try.
Just try. Keep working on what interests you.
Strengthening International Collaboration in Nanomedicine
Lucía Enríquez’s research stay exemplifies how international mobility enhances scientific innovation. By combining iPS cell technology from Kyoto with non-viral gene delivery systems and 3D bioprinting expertise from NANBIOSIS Unit 10, this collaboration advances personalized disease modeling for cystic fibrosis and strengthens Spain–Japan scientific ties.
Such synergies reflect the mission of NANBIOSIS: providing cutting-edge biomedical research infrastructures that empower researchers to push the boundaries of nanomedicine, gene delivery, and regenerative medicine.
What is NANBIOSIS?
The goal of NANBIOSIS is to provide comprehensive and integrated advanced solutions for companies and research institutions in biomedical applications. All of this is done through a single-entry point, involving the design and production of biomaterials, nanomaterials, and their nanoconjugates. This includes their characterization from physical-chemical, functional, toxicological, and biological perspectives (preclinical validation).
If you want to collaborate with us, visit our Order Request page.
In order to access our Cutting-Edge Biomedical Solutions with priority access, enter our Competitive Call here.
NANBIOSIS has worked with pharmaceutical companies of all sizes in the areas of drug delivery, biomaterials and regenerative medicine. Here are a few of them: