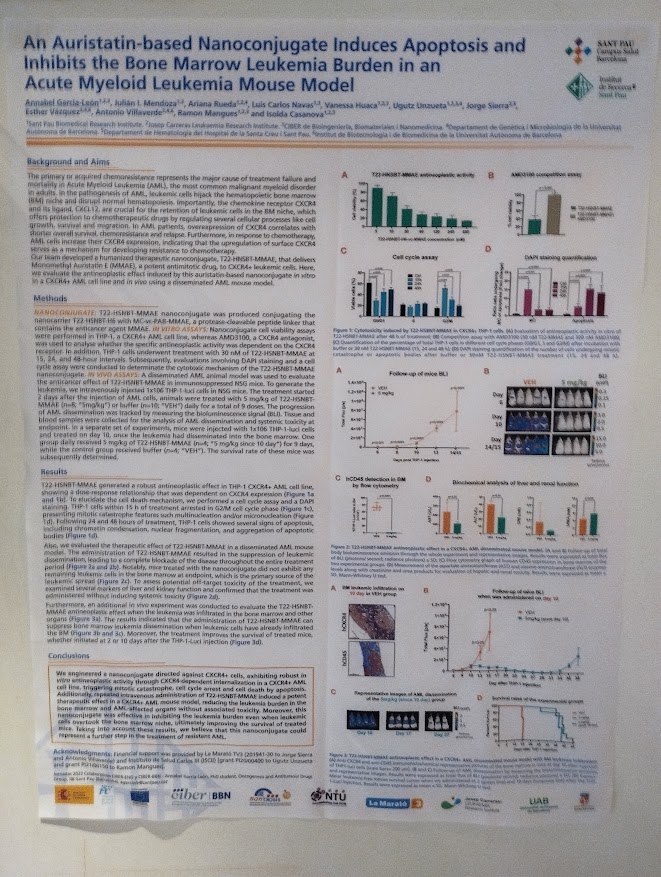
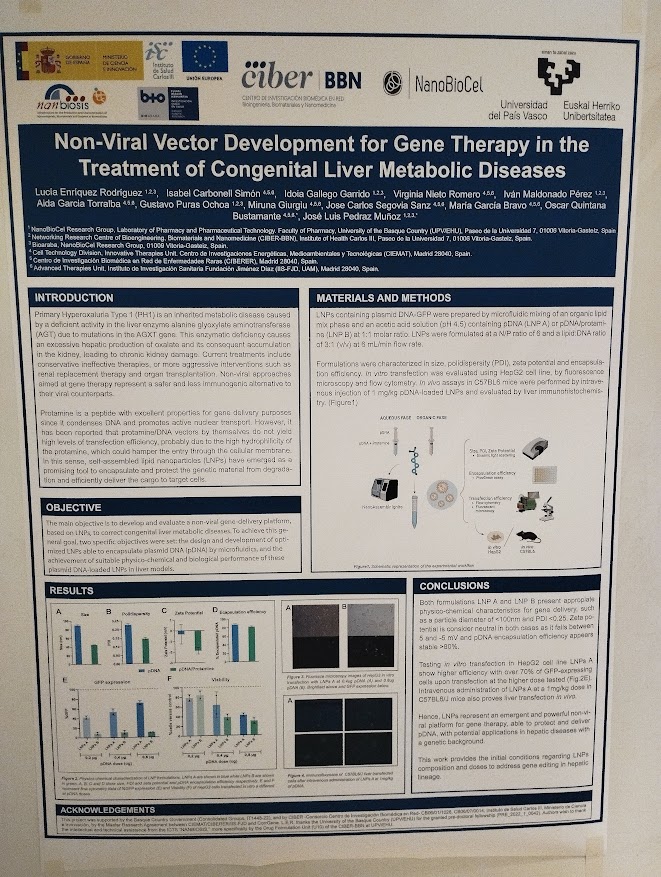
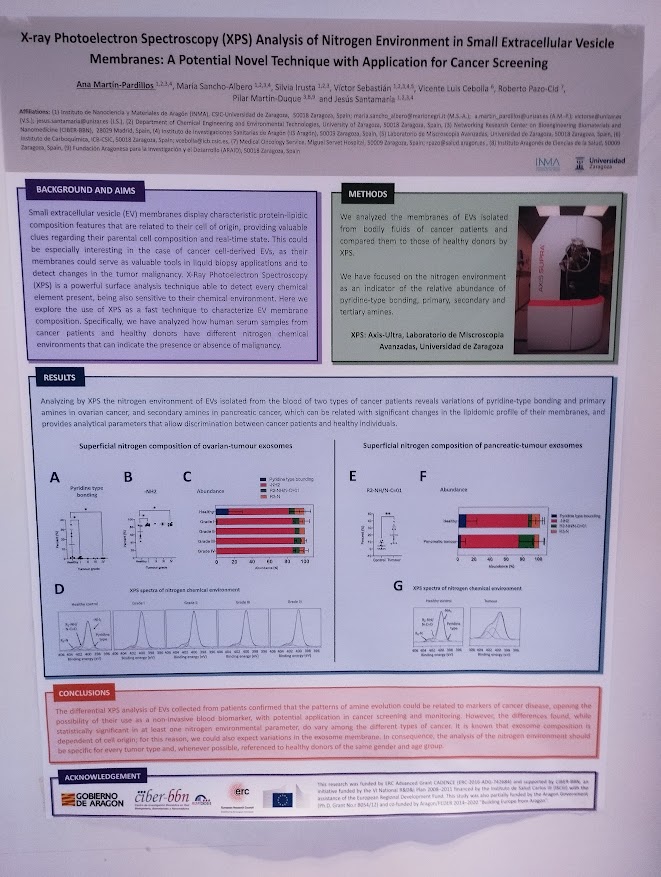
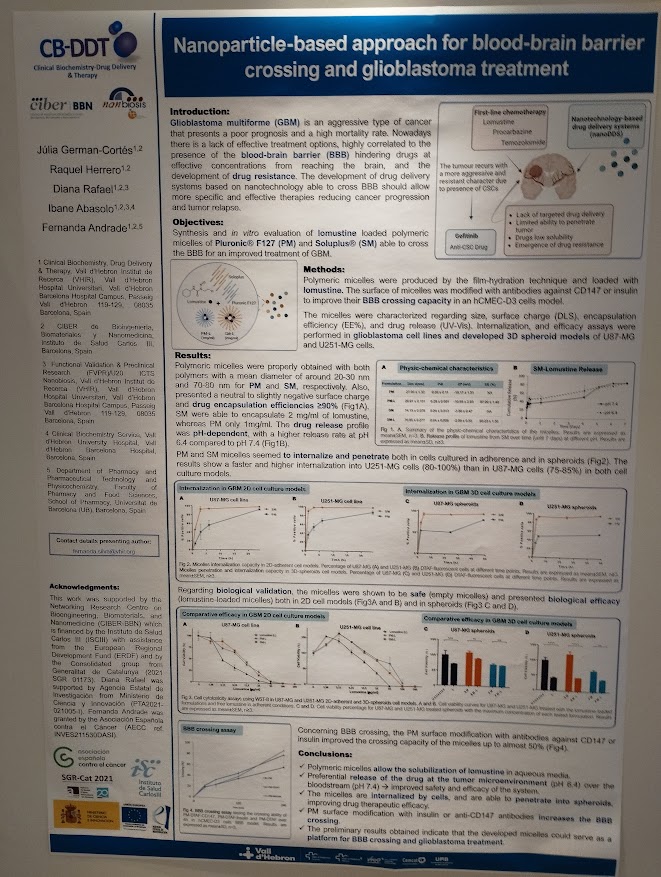
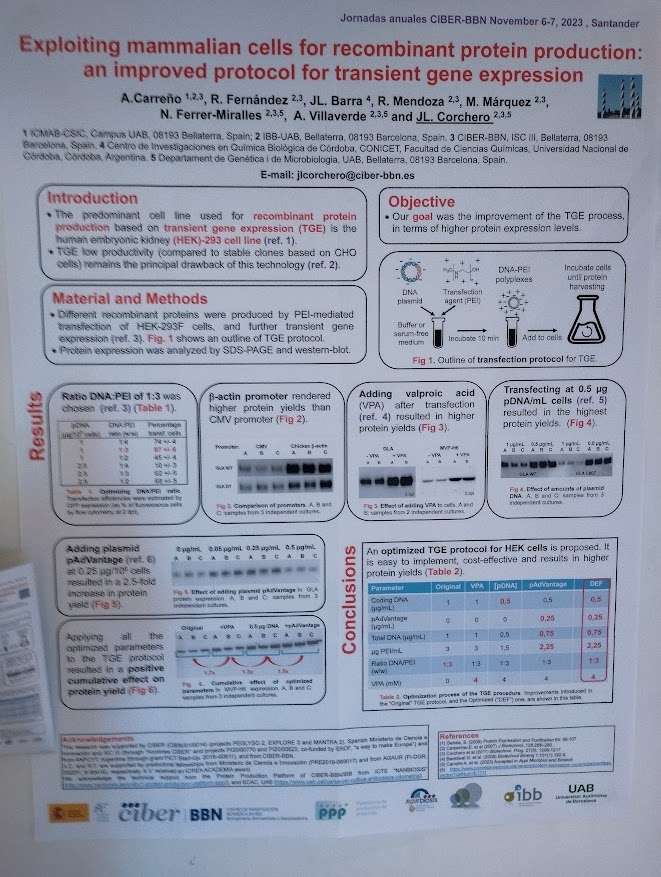
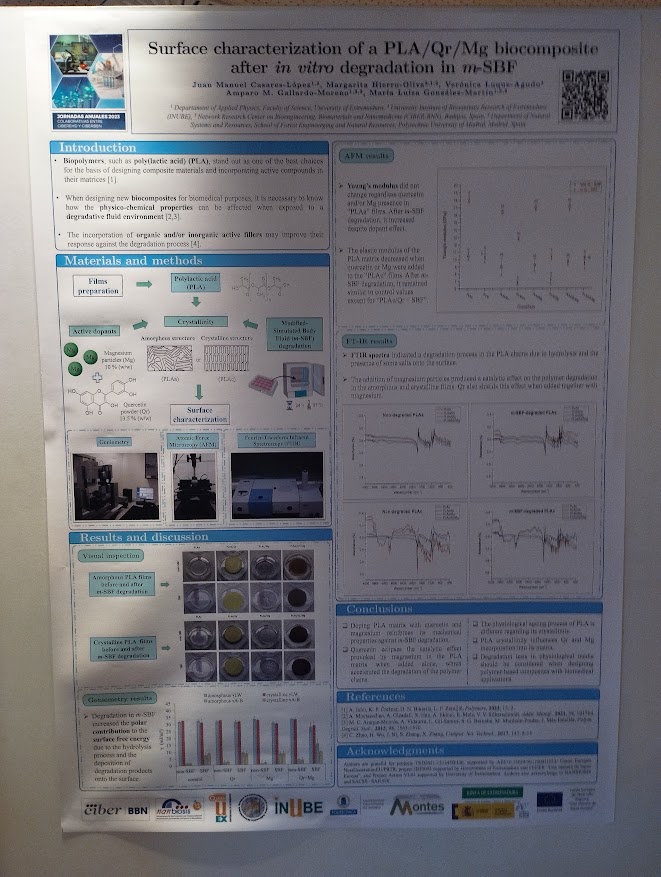
NANBIOSIS researchers contribute to the 2nd CSIC Rare Diseases Network Meeting in Madrid
NANBIOSIS researchers join the 2nd CSIC Rare Diseases Network Meeting in Madrid, strengthening interdisciplinary collaboration in rare disease research.
Madrid, February 2026 — Several researchers from NANBIOSIS and CIBER-BBN based at the IQAC-CSIC took part in the 2nd Meeting of the CSIC Rare Diseases Network, held in Madrid. The event brought together scientists, legal experts, patients, sociologists, and other key stakeholders to address rare diseases from a truly interdisciplinary perspective.
Among the participants were Miriam Royo, Scientific Director of Unit 3, Lluïsa Villaplana, researcher from Unit 2 and Ibane Abasolo, Strategy Advisor of NANBIOSIS. Their participation highlights the strong commitment within NANBIOSIS towards collaborative biomedical research and to advancing knowledge in rare diseases through cutting-edge infrastructures.
Interdisciplinary collaboration for Rare Diseases
Rare diseases affect millions of people worldwide, yet many remain underdiagnosed and lack effective treatments. The CSIC Rare Diseases Network meeting provided an enriching forum to exchange experiences not only among researchers, but also with professionals from the legal and social sciences, as well as patient representatives.
This multidisciplinary dialogue is essential to address the scientific, regulatory, and societal challenges associated with rare disease research, from biomarker discovery to preclinical validation and translational development.
NANBIOSIS Units supporting Rare Disease research
The participation of NANBIOSIS researchers reflects the strategic role of its Units in supporting high-impact biomedical research
Unit 3 – Synthesis of Peptides Unit
Unit 3, coordinated by the Multivalent Systems for Nanomedicine group at IQAC-CSIC, offers advanced services in peptide synthesis (from milligrams to grams), purification, characterization, and post-synthesis modifications, including conjugation to proteins and fluorescent labels.
With extensive expertise in biologically active peptide design and in their incorporation into therapeutic nanoconjugates, Unit 3 supports the development of innovative strategies for targeted therapies—an essential area for many rare diseases with limited treatment options.
Unit 2 – Custom Antibody Service (CAbS)
Unit 2 at IQAC-CSIC is equipped for the development and production of monoclonal and polyclonal antibodies, including hybridoma generation, antibody purification, immunoassays, and bioconjugate synthesis. The Unit operates under ISO9001 certification, ensuring standardized quality control procedures.
Its capabilities in antibody production and immunoreagent development are highly relevant for rare disease research, particularly in biomarker validation, diagnostic assay development, and targeted therapeutic approaches.
By participating in the 2nd CSIC Rare Diseases Network Meeting, NANBIOSIS researchers reinforce the infrastructure’s role as a key enabler of collaborative, translational, and interdisciplinary research in rare diseases—helping bridge the gap between fundamental science and clinical application.
What is NANBIOSIS?
The goal of NANBIOSIS is to provide comprehensive and integrated advanced solutions for companies and research institutions in biomedical applications. All of this is done through a single-entry point, involving the design and production of biomaterials, nanomaterials, and their nanoconjugates. This includes their characterization from physical-chemical, functional, toxicological, and biological perspectives (preclinical validation).
If you want to collaborate with us, visit our Order Request page.
In order to access our Cutting-Edge Biomedical Solutions with priority access, enter our Competitive Call here.
NANBIOSIS has worked with pharmaceutical companies of all sizes in the areas of drug delivery, biomaterials and regenerative medicine. Here are a few of them: