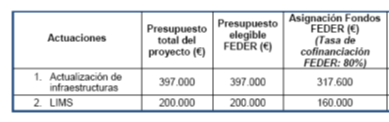
U8-S05. Deposition of thin films (Polyimide, Parylene) for the fabrication of flexible devices or encapsulation purposes (On-site&Remote) OUTSTANDING.
Deposition of thin films (Polyimide, Parylene) for the fabrication of flexible devices or encapsulation purposes (On-site&Remote) OUTSTANDING.
Deposition of Polyimide (PI-2611) via spin-coating for the fabrication of flexible free-standing structures.
Conformal deposition of Parylene (different types available)
Customer benefits
- Possibility to access to the PI deposition system in self-service mode to adapt the process to the customer needs.
- Different Parylene types available for deposition to match customer needs
Target customer
Research groups and SMEs
References
- Masvidal-Codina E, Illa X et al., Nature Materials 18 (2019) 280-288
- Bonaccini Calia et al., Nature Nanotechnology 17 (2022) 301-309
- Viana et al., Nature Nanotechnology (2024)