Wide representation of NANBIOSIS research in NALS 2022 and best Oral Presentation to Eloi Parladé
During the last 27-29 April, the 3rd International Conference on Nanomaterials Applied to Life Sciences 2022 (NALS 2022) has taken place in the Excellence Campus of Universidad de Cantabria in Santander (Spain), organized by the University of Cantabria and Institute Valdecilla-IDIVAL.
NALS 2022 has been a multidisciplinary conference series sharing new results and ideas in the fields of biosensors, lab on a chip, drug delivery nanopharmacy. nanobiotechnology, intelligent nanomaterials, magnetic materials, nanotoxicity, antimicrobials, novel applications of 3d bioprinting and nanoimaging.
NANBIOSIS has been represented at this edition by members of several of its units, among them we must highlight the set of four oral communication presented by members of the Nanobiotechnology group-Unit 1 of NANBIOSIS “Protein Production Platform (PPP)”, from CIBER-BBN and IIB-UAB the talks were delivered by:
Eloi Parladé: “Development of ion-dependent microscale secretory granules for nanomedical applications”
Carlos Martínez-Torró: “Design of a human GFP-like protein scaffold for targeted nanomedicines”
Eric Voltá Durán: “Antitumoral nanoparticles with multiple activities, a close reality”
Jan Atienza-Garriga: “Characterization of protein-only NPs containing amps and analysis of their protection with liposomes and micelles”
They summarized a wide area of the team activities on the design of protein-based protein materials for clinical uses, produced by means of diverse types of cell factories. In particular, antimicrobial peptides, cytotoxic proteins with antitumoral targeting and drug-carrying scaffold proteins are engineered to confer self-assembling properties as either microparticles or nanoparticles, that can be further functionalized with chemical drugs through covalent binding. Microparticles are of special interest as they can be used as slow drug delivery systems for nanostructured drugs upon subcutaneous administration. Alternatively, nanoparticles can be also presented as embedded in liposomes or other micellar structures that stabilize them for enhanced performance.
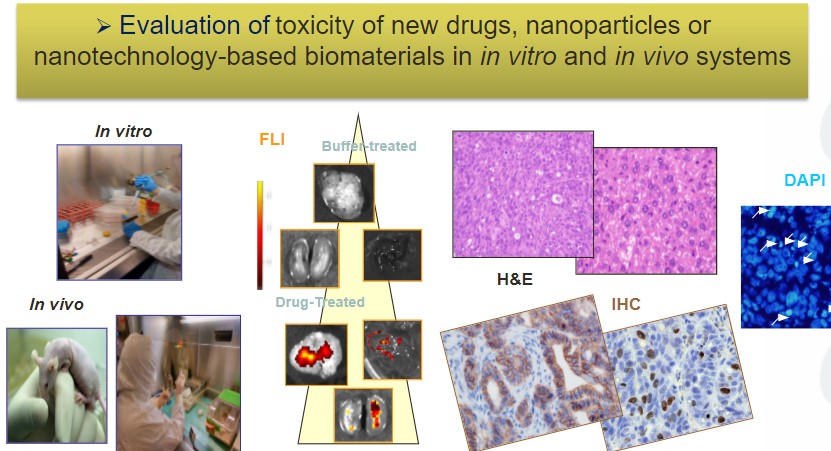
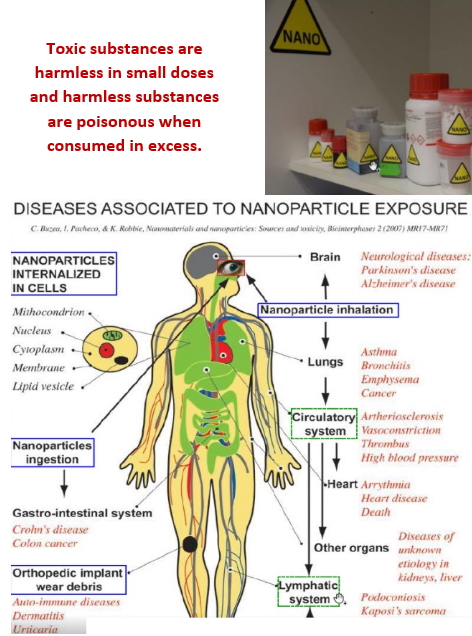
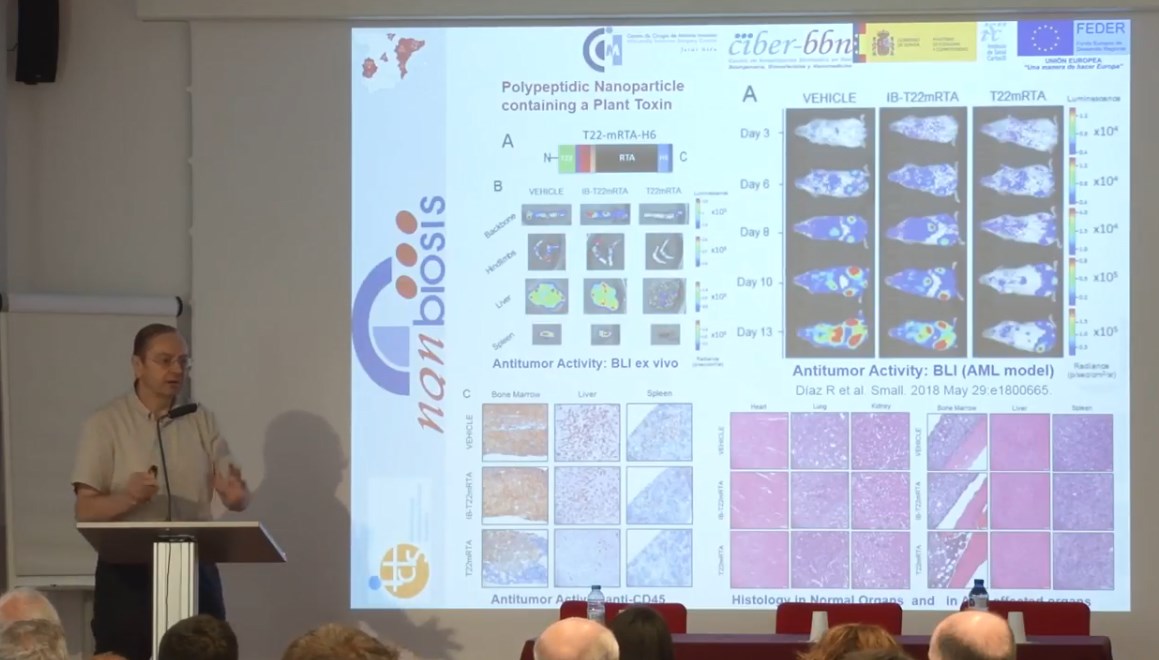
Three NANBIOSIS units supported the presented research, which has been executed in a highly cooperative way: namely U1 (Protein Production Platform), led by Tony Villaverde U18 (Nanotoxicology), led by Ramón Mangues and U29 (Oligonucleotide Synthesis Platform), led by Ramón Eritja.
Among all the excellent contributions by the team, it is worthy to stress that the prize for the best Oral Presentation was granted to Dr Eloi Parladé.
Other talk by researchers from NANBIOSIS were “Antioxidant-loaded polymeric NPs prepared by nano-emulsion templating for the management of neurological diseases” by Santiago Grijalvo, from NANBIOSIS U12 and “Exploiting GSH oxidation with nanocatalysts to promote cancer cell death” by Javier Bonet-Aletá from NANBIOSIS U9
On the other hand, Jesús Santamaría, Scientific Director of NANBIOSIS U9 was a Keynote Speaker in the Conference with the talk: “A change of paradigm in cancer therapy? Using catalysts to make drugs inside the tumor, rather than trying systemic chemotherapy”
NALS 2022 has been an excellent conference, with presentations covering a wide range of topics in nanomaterials for health, and a great opportunity for our researchers, especially for young’s, to let know their collaborative work, as well as make new connections on common research interests, thanks to the good socializing opportunities afforded by the scheduling of the organization conference.