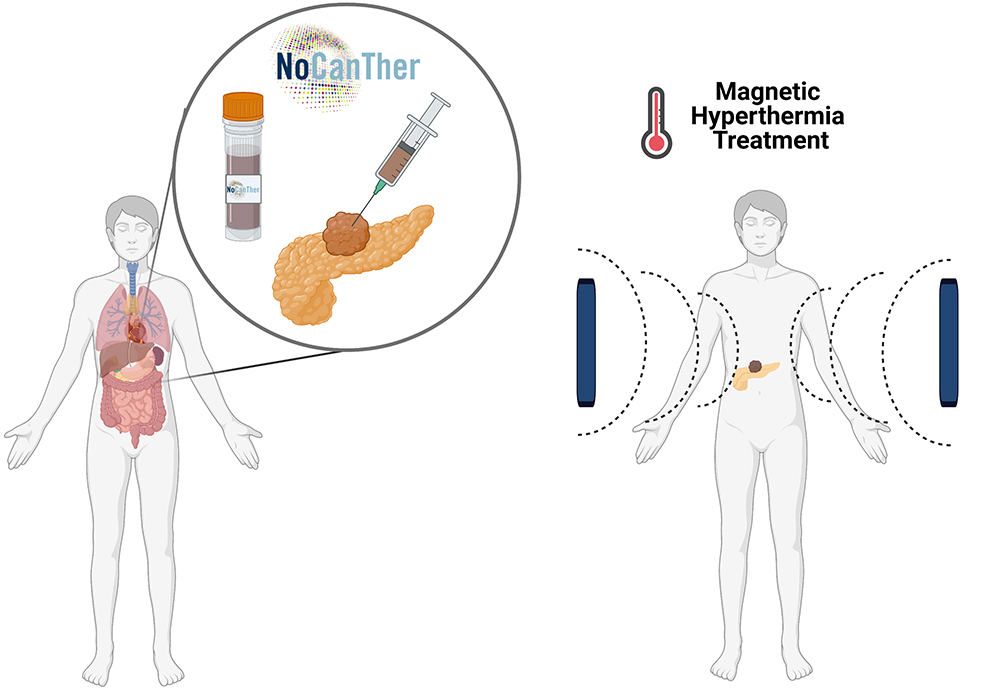
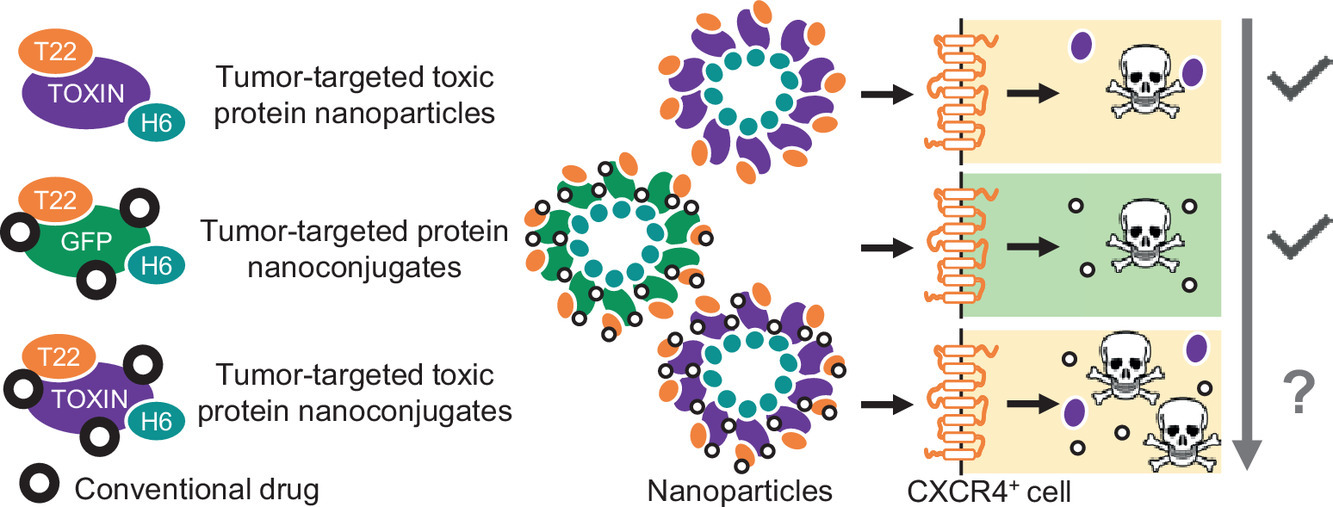
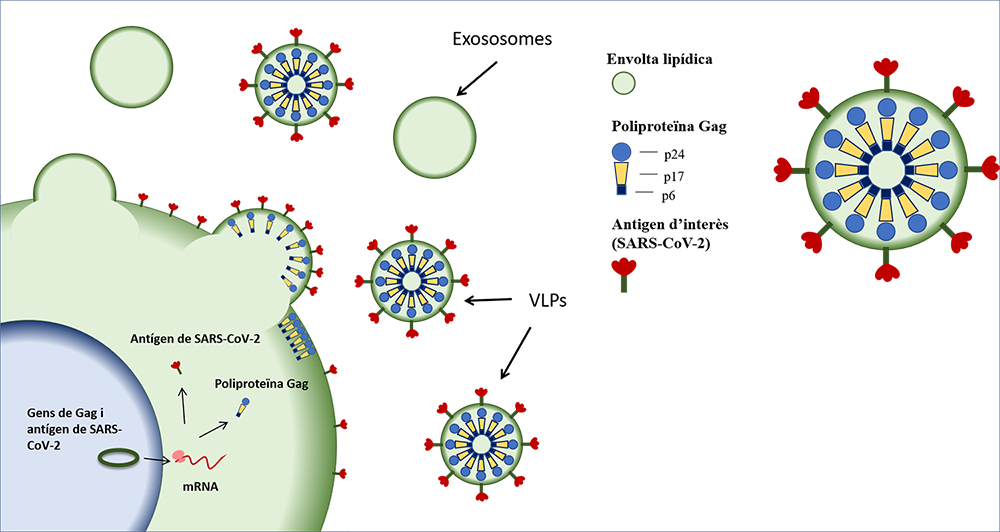
Nanoparticles to modulate topography and ligand distribution at the nanoscale: impact on cell behavior
Doctor Marc Martínez from the Nanomol-Bio group – NANBIOSIS U6 from CIBER-BBN at ICMAB-CSIC, defended his PhD thesis “Nanoparticles to modulate topography and ligand distribution at the nanoscale: impact on cell behavior” on 9 March 2023 at ICMAB.
he PhD thesis was supervised by Imma Ratera, Judith Guasch and Nora Ventosa from the Nanomol-Bio group at ICMAB-CSIC.
Ana Paula Candiota Silveira, Scientific Coordinator of NANBIOSIS U25 was part of the Committee that evaluated the Thesis tooghether with Jesús Martínez de la Fuente, Instituto de Nanociencia y Materiales de Aragón (INMA-CSIC) (President), and Anna Lagunas Targarona, Institut de Bioenginyeria de Catalunya (IBEC) (Vocal).
As Marc Martínez explained in an inteview to ICMAB “I produce nanoparticles in the lab and I use them to see how cells react to them. I work at the interface between cells and materials. My research can be applied to cell culture, which can be relevant for the development of new therapies for the regeneration of organs or for building implants to replace missing organs and body parts”.
Doctor Marc Martínez’s PhD thesis was part of the PhD Programme in Biochemistry, Molecular Biology and Biomedicine from the Universitat Autònoma de Barcelona (UAB).
Further information at ICMAB webpage