
NANBIOSIS Unit 12 showcased advanced colloidal characterization technologies at the 54 CED Annual Meeting, highlighting their role in product innovation.
Barcelona, March 2026 — Last March 11–12, NANBIOSIS Unit 12 actively participated in the 54 CED Annual Meeting, a key international forum bringing together industry leaders and scientific experts in detergency, cosmetics, and colloidal systems.
The event provided a valuable platform for knowledge exchange and collaboration between academia and industry, reinforcing the importance of advanced characterization techniques in the development and optimization of innovative products.
Advancing Colloidal Science for Industry Applications
During the meeting, NANBIOSIS Unit 12 highlighted its cutting-edge capabilities in the comprehensive characterization of colloidal systems. The Unit emphasized the critical role of particle analysis in ensuring product performance, stability, and quality control across sectors such as cosmetics, pharmaceuticals, and detergents.
Among the featured contributions were:
- Oral presentation: “Particle size as a key parameter in the characterization of colloids”, delivered by Susana Vílchez
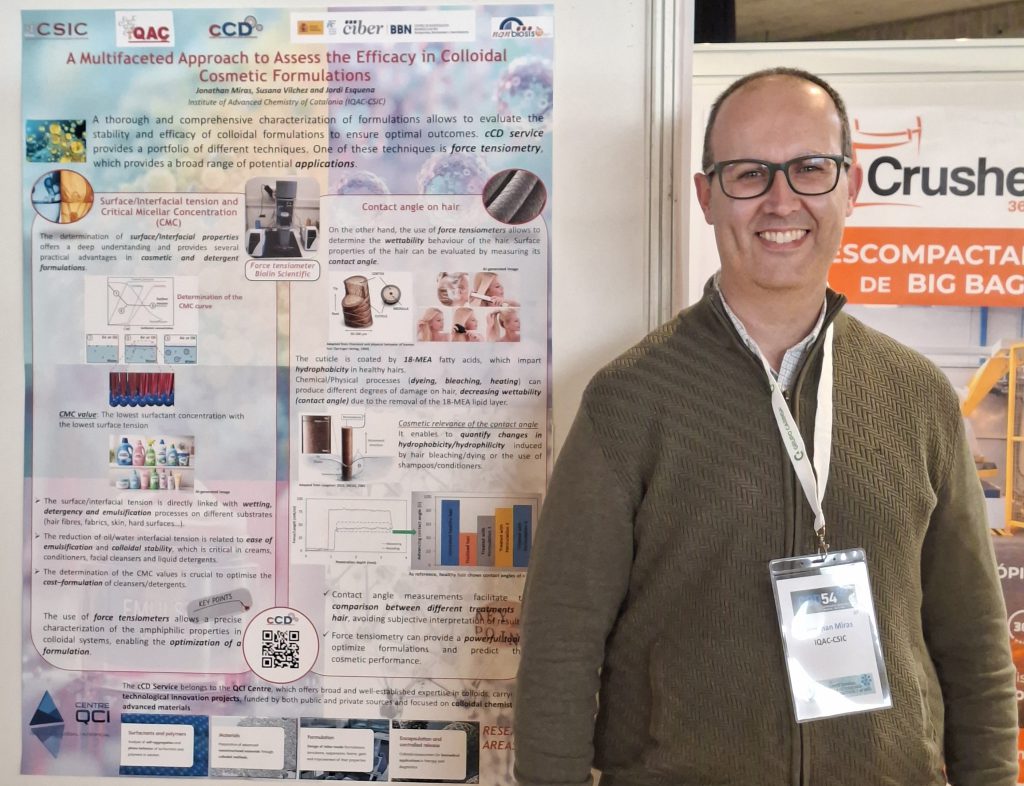
- Poster presentation: “A Multifaceted Approach to Assess the Efficacy in Colloidal Cosmetics Formulations”, presented by Jonathan Miras

These contributions showcased how advanced analytical techniques can provide deeper insights into colloidal formulations, ultimately supporting more efficient product development and improved consumer outcomes.
About NANBIOSIS Unit 12
NANBIOSIS Unit 12 (Nanostructured Liquid Characterization) is hosted at the Institute of Advanced Chemistry of Catalonia (IQAC-CSIC), part of the Spanish National Research Council (CSIC), in Barcelona.
The Unit is coordinated by the Colloid and Interfacial Chemistry Group and led by Scientific Director Prof. Carlos Rodríguez, with Jordi Esquena as Scientific Coordinator.
Unit 12 specializes in the characterization of nanostructures in liquid systems and interfaces, including micelles, liposomes, nanoemulsions, and nanoparticles. Its state-of-the-art infrastructure enables detailed analysis of key parameters such as:
- Particle size distribution and zeta potential
- Critical micelle concentration (CMC)
- Surface and interfacial tension
- Colloidal stability and solubilization capacity
- Rheological properties (viscosity, shear, and elastic moduli)
These capabilities make Unit 12 a strategic partner for both academic and industrial stakeholders seeking advanced solutions in nanomedicine, cosmetics, and materials science.
Strengthening Industry Collaboration Through NANBIOSIS
Participation in events like the CED Annual Meeting highlights NANBIOSIS’ commitment to bridging scientific excellence and industrial innovation. By showcasing its technological capabilities and engaging with industry professionals, Unit 12 continues to foster collaborations that accelerate the translation of research into real-world applications.
More information about the event here.
What is NANBIOSIS?
The goal of NANBIOSIS is to provide comprehensive and integrated advanced solutions for companies and research institutions in biomedical applications. All of this is done through a single-entry point, involving the design and production of biomaterials, nanomaterials, and their nanoconjugates. This includes their characterization from physical-chemical, functional, toxicological, and biological perspectives (preclinical validation).
If you want to collaborate with us, visit our Order Request page.
Leading scientists
The main value of NANBIOSIS is our highly qualified and experienced academic scientists, working in public institutions, renowned universities and other research institutes.
Custom solutions
Designed for either scientific collaboration or the private industry, we adapt our services to your needs, filling the gaps and paving the way towards the next breakthrough.

Cutting-Edge facilities
Publicly funded, with the most advanced equipment, offering a wide variety of services from synthesis of nanoparticles and medical devices, including up to preclinical trials.
Standards of quality
Our services have standards of quality required in the pharmaceutical, biotech and medtech sectors, from Good Practices to ISO certifications.
In order to access our Cutting-Edge Biomedical Solutions with priority access, enter our Competitive Call here.
NANBIOSIS has worked with pharmaceutical companies of all sizes in the areas of drug delivery, biomaterials and regenerative medicine. Here are a few of them:









