Reversing pulmonary fibrosis has been achieved in murine models with peptide-loaded nanoparticles
A new study with participation of NANBIOSIS Unit 10 Drug Formulation unit of CIBER-BBN and UPV/EHU, has succeeded in preventing and reversing pulmonary fibrosis in experimental models with PLGA / PEI nanoparticles loaded with the GSE4 peptide. These results are the first step to study its possible antifibrotic effect in patients with idiopathic pulmonary fibrosis and other progressive fibrosing diffuse interstitial lung diseases that present altered telomere function.
NANBIOSIS unit 10 has been responsible for the design and manufacture of biodegradable nanoparticles loaded with the peptide.
The work is led by Rosario Perona and Leandro Sastre, researchers of CIBERER at the Biomedical Research Institute (CSIC/UAM), and Maria Molina-Molina, researcher at CIBERES and the Department of Pulmonology at the Bellvitge University Hospital. In addition, Guillermo Güenechea, from CIBERER at CIEMAT-IIS / FJD, and José Luis Pedraz, from CIBER-BBN and the University of the Basque Country have collaborated.
Idiopathic pulmonary fibrosis is a fatal fibrotic disease associated with aging with a median survival of 2 to 5 years. The disease usually appears around the age of 60, but there are familiar forms that can manifest at younger ages. Fibrosis appears as an aberrant healing response after alveolar damage and is characterized by the appearance of abnormal lung scarring, in which the restoration of epithelial integrity and tissue function is compromised. The mechanisms responsible for defective repair and regeneration are not well understood. However, recent studies suggest a possible role of accelerated aging and telomere shortening in the onset of the disease, mainly in familial forms.
The GSE4 peptide, of 11 amino acids, corresponds to an internal sequence of dyskerin, a protein that is part of the telomerase complex. GSE4 has protective effects on the cells of patients with the rare diseases dyskeratosis congenita and ataxia-telangiectasia, as it increases telomerase activity and cell growth and reduces DNA damage, oxidative stress and cell senescence.
The authors of the study, published in The FASEB Journal, used rat lung alveolar cells treated with a profibrotic agent, bleomycin, as a model. Expression of the GSE4 peptide or treatment with GSE4-PLGA / PEI nanoparticles causes a reversal of the inflammatory and fibrotic phenotype. There is an increase in telomerase activity, a decrease in DNA damage, and a decrease in inflammation and cell death. Furthermore, these cells show an inhibition in fibrosis.
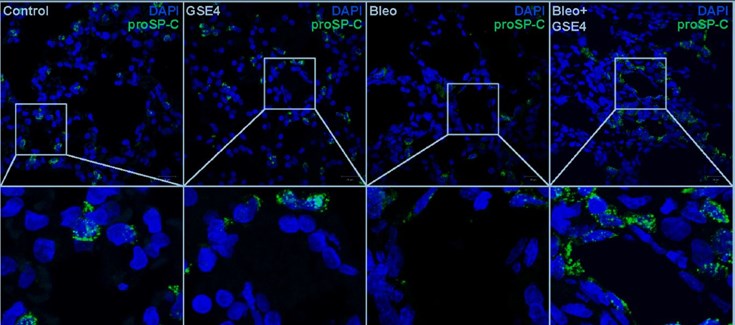
In a rat model with bleomycin-induced pulmonary fibrosis, treatment with GSE4-PLGA / PEI nanoparticles has also increased telomerase activity and decreased DNA damage in alveolar cells. In both preventive and therapeutic protocols, GSE4-PLGA / PEI nanoparticles have prevented and attenuated lung damage and inhibited fibrosis. Histological analysis of the lungs of rats treated with bleomycin and GSE4-PLGA / PEI nanoparticles shows less fibrosis and inflammation and greater regeneration of the alveolar tissue, indicating the therapeutic efficacy of GSE4-loaded nanoparticles in this experimental pulmonary fibrosis model. .
This study supports the possibility of further investigating a potential curative treatment for patients with pulmonary fibrosis.
Reference article:
Pintado-Berninches, L., Montes-Worboys, A., Manguan-García, C., et al. “GSE4-loaded nanoparticles a potential therapy for lung fibrosis that enhances pneumocyte growth, reduces apoptosis and DNA damage.” The FASEB Journal. 2021; 35: e21422. DOI: 10.1096/fj.202001160RR
(*) Explanation of the figure: Treatment with the profibrotic agent bleomycin decreases the number of cells that express the alveolar marker proSP-C (Bleo). Inoculation of GSE4-loaded nanoparticles into bleomycin-treated rats increases the number of cells expressing the marker, indicating alveolar regeneration.