NANBIOSIS advances targeted nanomedicine for Acute Myeloid Leukemia on World Cancer Day
NANBIOSIS Unit 18 contributes to two studies advancing CXCR4-targeted nanoconjugates for acute myeloid leukemia, highlighted on World Cancer Day.
On the occasion of World Cancer Day, NANBIOSIS highlights the recent publication of two scientific articles advancing a novel line of research focused on the development of targeted nanoconjugates for the treatment of acute myeloid leukemia (AML). Both studies include the active participation of NANBIOSIS Unit 18, reinforcing the Unit’s role in the preclinical development and evaluation of innovative cancer nanomedicines.
Optimizing conjugation strategies to enhance antitumor efficacy
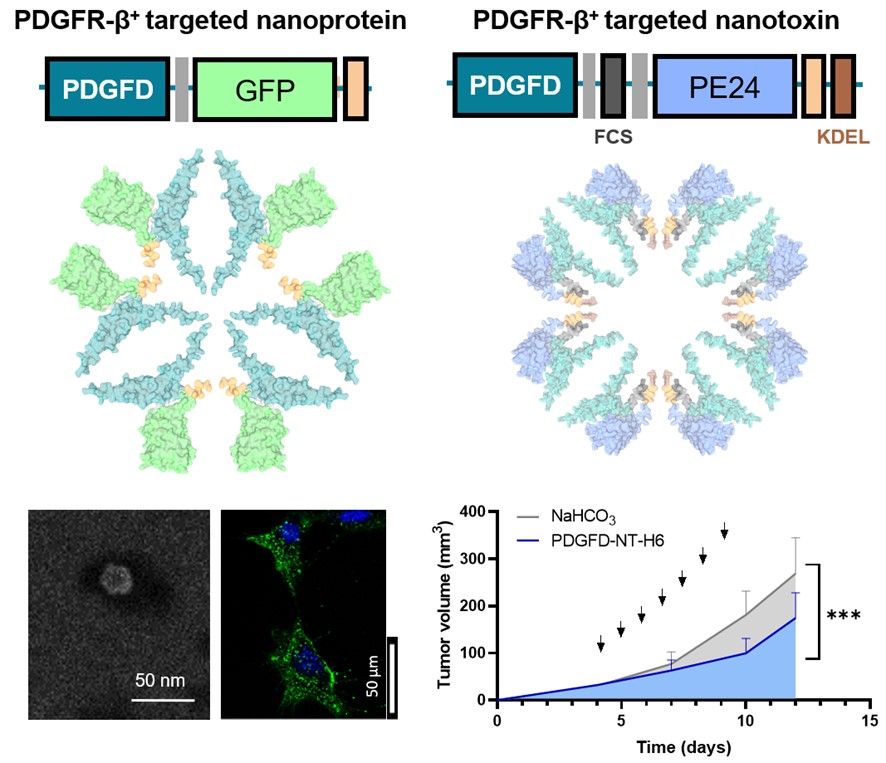
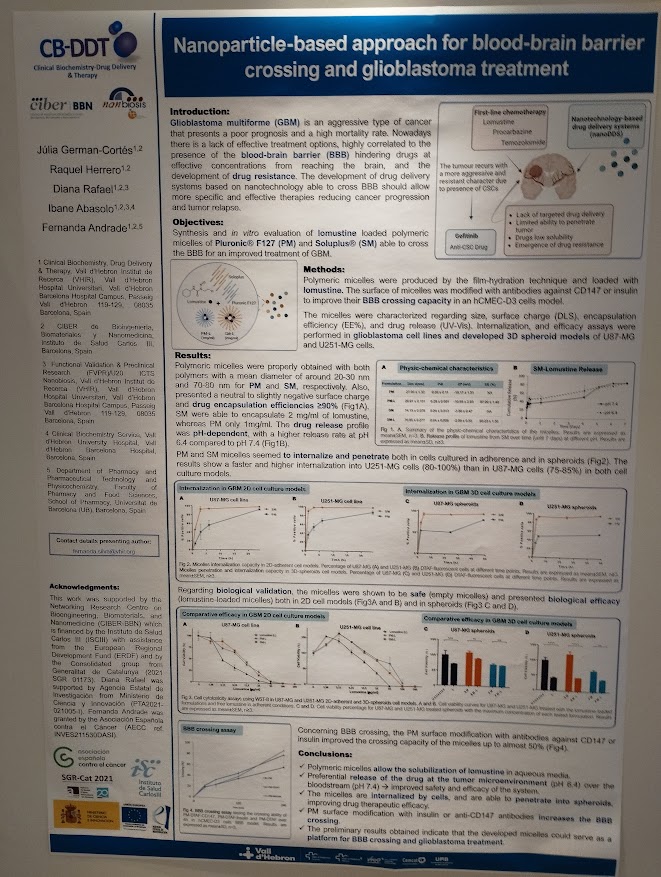
The first study, “Conjugation strategy shapes antitumor efficacy and enables dose-sparing in non-antibody protein nanoconjugates”, addresses a key challenge in cancer nanomedicine: improving therapeutic selectivity while minimizing systemic toxicity. While antibody–drug conjugates (ADCs) are considered the gold standard in targeted therapies, their clinical limitations have driven the search for alternative approaches.
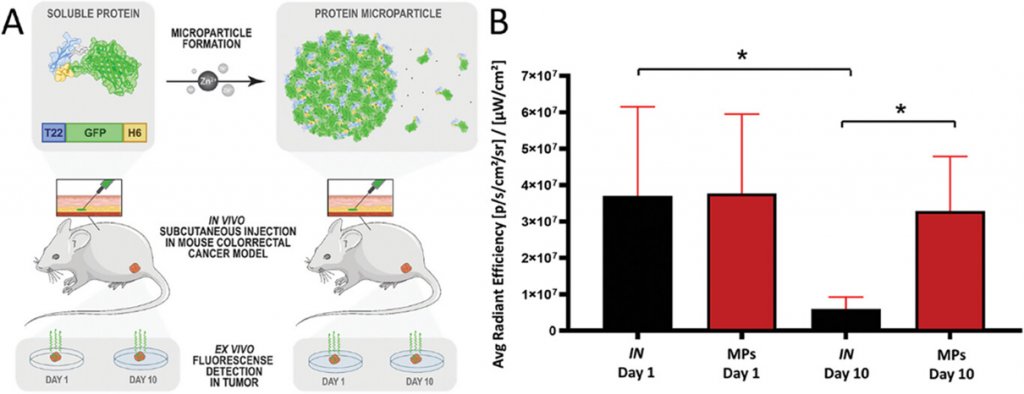
In this work, researchers evaluated how site-specific conjugation strategies influence the biodistribution and antitumor efficacy of a CXCR4-targeted multivalent protein nanocarrier loaded with the cytotoxic drug monomethyl auristatin E (MMAE). The results demonstrated that precise payload positioning significantly impacts therapeutic performance. Notably, a site-specific design achieved comparable tumor control with a four-fold lower drug load, highlighting the potential of dose-sparing strategies in next-generation protein-based nanomedicines.
Precision targeting of CXCR4⁺ leukemia cells in AML models
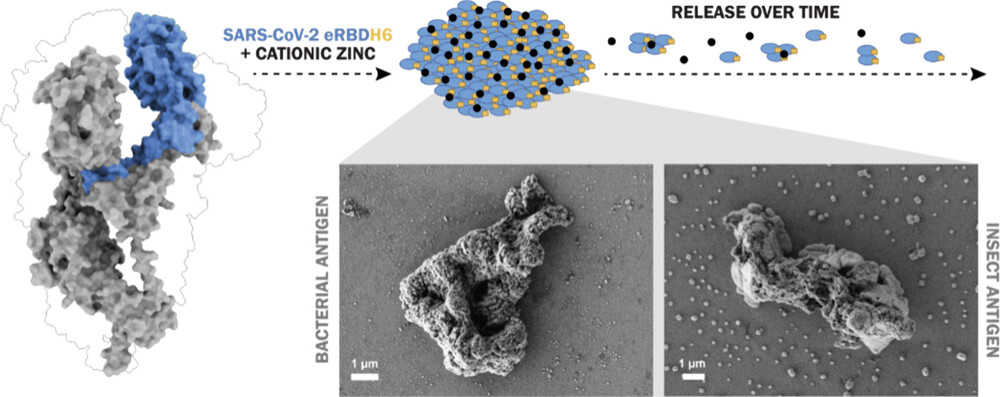
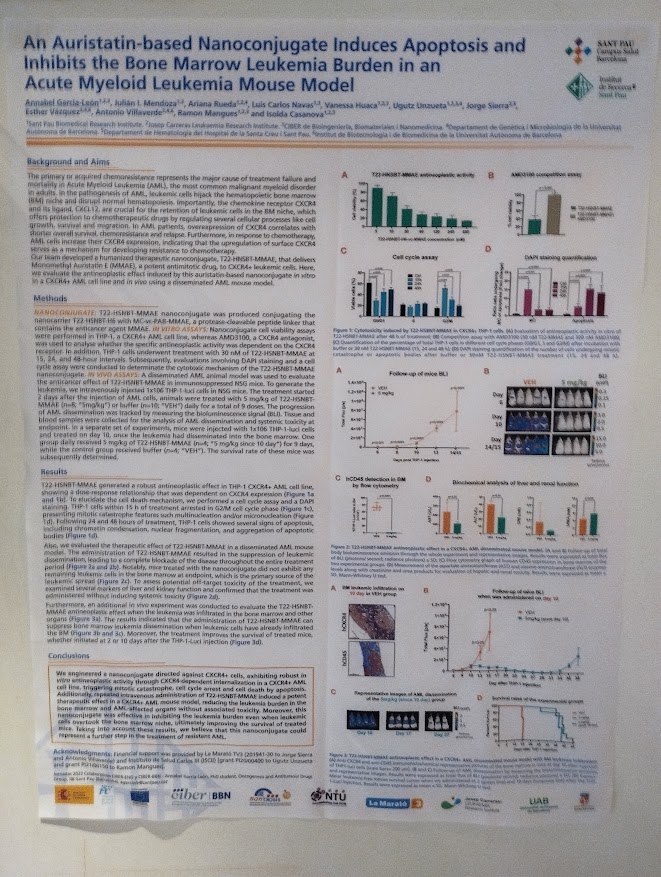
The second publication, “Precision targeting of CXCR4+ leukemia cells by a humanized MMAE-nanoconjugate in an AML mouse model”, focuses on overcoming chemoresistance and relapse in AML, which are largely driven by therapy-resistant leukemic stem cells in the bone marrow.
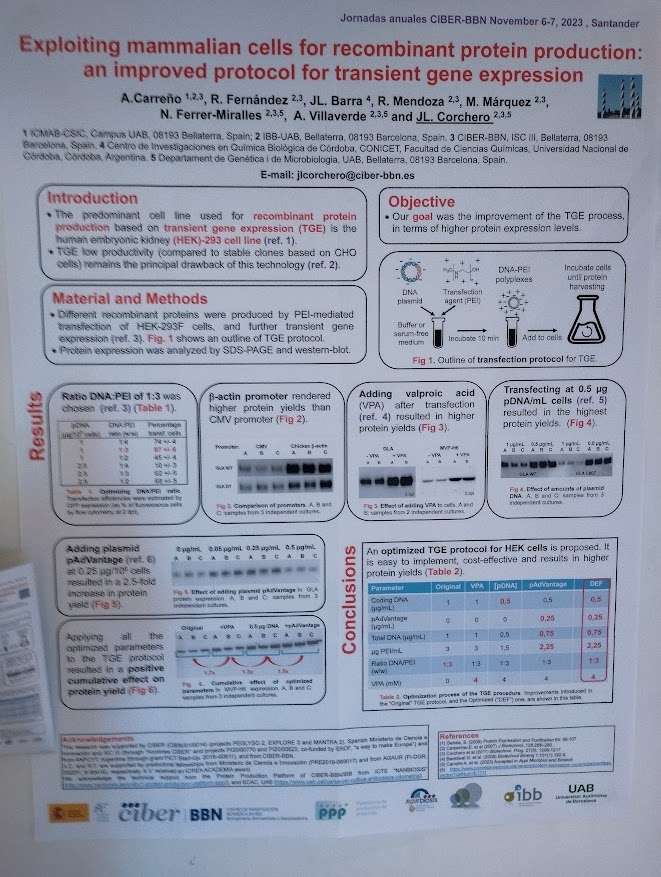
The study describes the development of T22-HSNBT-H6-MMAE, a humanized nanoconjugate designed to selectively target CXCR4-overexpressing leukemia cells, a biomarker associated with poor prognosis in approximately 50% of AML patients. In vitro and in vivo experiments demonstrated potent antileukemic activity, including complete suppression of leukemic dissemination and prolonged survival in a disseminated AML mouse model, without systemic toxicity. Importantly, the nanoconjugate showed selectivity for malignant cells while sparing healthy bone marrow and peripheral blood cells.
The role of NANBIOSIS Unit 18
NANBIOSIS Unit 18, located at the Hospital de la Santa Creu i Sant Pau (Barcelona) and coordinated by Dr. Ramón Mangues, specializes in nanotoxicology and preclinical safety assessment. The Unit evaluates the toxicity and tolerability of new drugs, nanoparticles and nanotechnology-based biomaterials using in vitro and in vivo models, supporting the optimization of lead compounds with higher probabilities of clinical success.
With advanced facilities for cell culture, flow cytometry, confocal microscopy and in vivo experimentation in small animal models, Unit 18 plays a key role in ensuring the safety, efficacy and translational potential of innovative nanomedicine approaches.
Together, these two studies underscore the contribution of NANBIOSIS to cutting-edge cancer research and highlight the promise of CXCR4-targeted nanoconjugates as a new therapeutic strategy for patients with acute myeloid leukemia.
What is NANBIOSIS?
The goal of NANBIOSIS is to provide comprehensive and integrated advanced solutions for companies and research institutions in biomedical applications. All of this is done through a single-entry point, involving the design and production of biomaterials, nanomaterials, and their nanoconjugates. This includes their characterization from physical-chemical, functional, toxicological, and biological perspectives (preclinical validation).
If you want to collaborate with us, visit our Order Request page.
In order to access our Cutting-Edge Biomedical Solutions with priority access, enter our Competitive Call here.
NANBIOSIS has worked with pharmaceutical companies of all sizes in the areas of drug delivery, biomaterials and regenerative medicine. Here are a few of them: